
(a)
Interpretation:
The set of reagents satisfying the given conditions used for the synthesis of the given
Concept introduction:
An
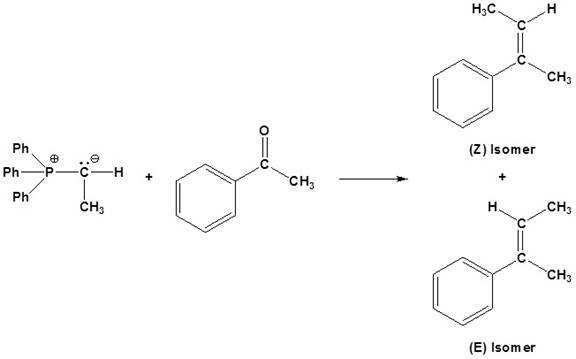
(b)
Interpretation:
The
Concept introduction:
An aldehyde or a ketone reacts with phosphonium ylide to form an alkene. This reaction is known as wittig reaction.
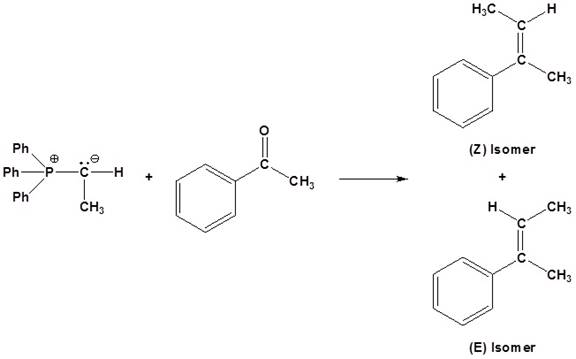
(c)
Interpretation:
The reagents used for the synthesis of given set of alkenes should be determined.
Concept introduction:
An aldehyde or a ketone reacts with phosphonium ylide to form an alkene. This reaction is known as wittig reaction.
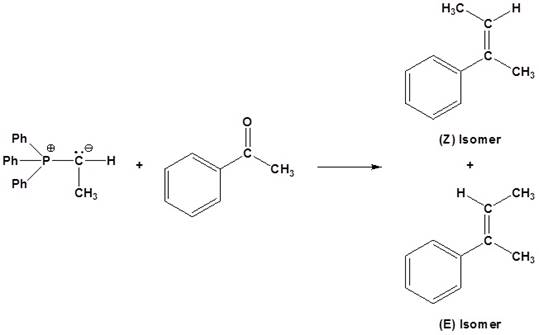
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
- Explain why Jone's oxidation of 4-methyl-2-pentanol is faster than that of 3,3-dimethyl-2-pentanol.arrow_forwardWhat aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the attached compounds?arrow_forwardA) Draw the final major product(s) for the following reaction. Please include proper stereochemistry B) propose an alternative carbonyl compound and ylide that could be used to make the same product C) of the reagents in a) or b) which is the more effective route for obtaining the final product and why? D) provide a reaction (mechanism not required) for making the ylide given in part Aarrow_forward
- explain why carbonyl compounds are so attractive to both nucleophiles and electrophilesarrow_forwardWhat aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the attached compound?arrow_forwardFill in the reagents a–c that are missing from the following scheme. Specify if any reaction conditions required in each step:arrow_forward
- Explain why a nucleophilic reagent such as ethoxide adds to an alkyne more easily than it adds to an alkenearrow_forwardThe anticoagulant Warfarin can be synthesized by a conjugate addition (Michael addition) reaction as shown below: a) Propose a mechanism for the reaction starting from the enolate shown below. You can assume water is present for any proton transfers needed.b) We saw that the keto tautomer is normally preferred over the enol tautomer, however, the enol tautomer is present in the final product (Warfarin) and that is the more stable tautomer in this case. Why do you think the enol tautomer is more stable? Hint: consider the molecule as a whole.arrow_forwardWhat is the likely mechanism of nucleophilic substitution for each alkyl halide?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

