
a.
Interpretation:
Name the given compound.
Concept introduction:
The nomenclature of
Naming Aldehydes:
Aldehydes have at least one hydrogen attached to the carbonyl carbon atom.
The IUPAC naming of an aldehydes is obtained by replacing the final "e" on the name of the parent hydrocarbon with "al".
The common name of the aldehyde is substituted for "oic acid" at the end of parent hydrocarbon chain.
For example:
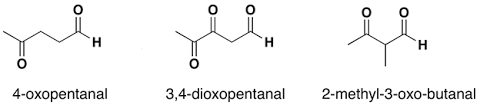
Naming Ketones:
Carbonyl carbon atom attached to the two oxygen atoms.
The IUPAC name of a ketones are obtained by replacing the "e" on the end of the parent hydrocarbon with "one".
Only few ketones have common name.
For example:
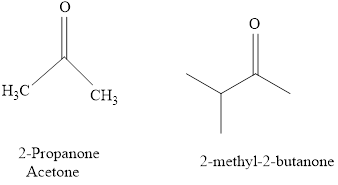
b.
Interpretation:
Name the given compound.
Concept introduction:
Naming Aldehydes:
Aldehydes have at least one hydrogen attached to the carbonyl carbon atom.
The IUPAC naming of an aldehydes is obtained by replacing the final "e" on the name of the parent hydrocarbon with "al".
The common name of the aldehyde is substituted for "oic acid" at the end of parent hydrocarbon chain.
For example:

Naming Ketones:
Carbonyl carbon atom attached to the two oxygen atoms.
The IUPAC name of a ketones are obtained by replacing the "e" on the end of the parent hydrocarbon with "one".
Only few ketones have common name.
For example:

c.
Interpretation:
Name the given compound.
Concept introduction:
Naming Aldehydes:
Aldehydes have at least one hydrogen attached to the carbonyl carbon atom.
The IUPAC naming of an aldehydes is obtained by replacing the final "e" on the name of the parent hydrocarbon with "al".
The common name of the aldehyde is substituted for "oic acid" at the end of parent hydrocarbon chain.
For example:

Naming Ketones:
Carbonyl carbon atom attached to the two oxygen atoms.
The IUPAC name of a ketones are obtained by replacing the "e" on the end of the parent hydrocarbon with "one".
Only few ketones have common name.
For example:

d.
Interpretation:
Name the given compound.
Concept introduction:
Naming Aldehydes:
Aldehydes have at least one hydrogen attached to the carbonyl carbon atom.
The IUPAC naming of an aldehydes is obtained by replacing the final "e" on the name of the parent hydrocarbon with "al".
The common name of the aldehyde is substituted for "oic acid" at the end of parent hydrocarbon chain.
For example:

Naming Ketones:
Carbonyl carbon atom attached to the two oxygen atoms.
The IUPAC name of a ketones are obtained by replacing the "e" on the end of the parent hydrocarbon with "one".
Only few ketones have common name.
For example:

Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





