
(a)
Interpretation: The name of the following compound should be determined:
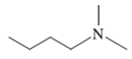
Concept Introduction: The group that contains a nitrogen atom attached to one or more carbon atoms is said to be an amine group, general representation of an amine group is R1R2R3-N.
The common name of
(b)
Interpretation: The name of the following compound should be determined:
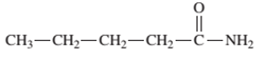
Concept Introduction: An organic compound in which carboxy
The reaction which results in the formation of amide along with water on heating acids with amine or ammonia is said to be amidation.
So, in order to give the IUPAC name to the amides, the rules for naming carboxylic acid is followed and -oic acid of the carboxylic acid is replaced by amide.
In order to give the name to the amide group, the following steps are followed:
- The parent (longest)
alkane chain is named as for carboxylic acids. - The -oic acid in the name is changed to -amide.
- The numbering of the chain is done in such a way that amide group and substituents gets the smaller number.
- N-alkyl is used to show each alkyl group bonded to -N atom in the name for secondary and tertiary amides.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
In order to write the common name of the amides, the common of acids are written from which the amide has been formed by replacing -oic acid in name from -amide.
(c)
Interpretation: The name of the following compound should be determined:
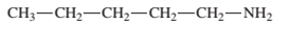
Concept Introduction: The group that contains a nitrogen atom attached to one or more carbon atoms is said to be an amine group, general representation of an amine group is R1R2R3-N.
The common name of amines are derived by naming the alkyl groups bonded to nitrogen atom in alphabetical order and then ending with word -amine.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
- (12.9) Which of the following has the highest boiling point? O Both H₂S and H₂O have the same boiling point that is higher than the boiling point of H₂Se. O H₂Se O H₂O O H₂Sarrow_forward12.58 Draw the condensed structural or line-angle formula, if cyclic, for each of the following: (12.3) a. formaldehyde c. 3-methyl-2-hexanone d. 3,5-dimethylhexanal b. 2-chlorobutanalarrow_forwardSimple alcohols (just containing C's, H's and an OH, where the H on the O is the acidic H) have a pKa similar to water (15.7) hydronium ion (-2)arrow_forward
- 12.42 Which of the following will give a positive Tollens' test? (12.4) 1-propanol b. 2-propanol c. hexanal a.arrow_forwardPage 1 > of 5 3. The oil painting "Irises" by Vincent van Gogh measures 28 inches high by 37 inches wide. (2.3, 2.6) a. What is its height in centimeters (cm)? b. What is its width in centimeters (cm)? C. Calculate the area of the painting, in square centimeters and square inches, to the correct number of significant figures. (A = w * h)arrow_forwardHelp me with all please !!! (1 to 5)arrow_forward
- Rank the following substances in order of increasing acidity: Which, if any, of the four compounds is a strong enough acid to react almost completely OH CHyCH3 CHyČCH,ČCH, CH,COH Acetone Pentane-2,4-dione Phenol Acetic acid with NaOH? (pK, 19.3) (pK, = 9) (pK, = 9.9) (pK,4.76) (The pK, of H20 is 15.74.)arrow_forwardwhat starting materials and reagents could form the indicated product ? Fill in the reaction, acco unt for H indicated as being from the reaction 1.2.3.4.5arrow_forwardGive only typing answer with explanation and conclusion Which of the following statements is/are incorrect? (6,25 p) I- In order for an organic compound to be a dyestuff, it must be chromor and auxochrochrome. II- Hydroxyl or amino groups can be given as examples of chromophore groups III- Decrease of chromophore groups in a molecule causes color deepening. A. b) II, III B. a) I only C. d) II only D. c) III onlyarrow_forward
- 12:46 16.48 Give the IUPAC and common names, if any, for each of the following compounds: (16.1, 16.4) CH3 a. CH3-CH-CH2-CH2-c-OH hey C- HO- b. Cl d. CH3-CH2–CH2-C-0–CH3 CH3 C-0-CH,-CH3 e. CH3-CH – CH2 CH3 OH f. CH3 CH- CH2 -СН-С-ОНarrow_forwardWhat is the value of the exponent in the answer? (104) (10 2) O -2 O -8 O 6 O 2arrow_forward7. What is the ordered pair corresponding to the point marked "A" in this А. (4, 14.0) В. (4, 10.4) С. (4, 5.1) D. (4, 4.1) graph? 10 10 10° 1 3 4 6 9. 10 8. How much kinetic energy does an electron (m = 9.1x10 kg) have if it is A. 1.0x1014 B. 1.4x10" J C. 2.7x102 J D. 6.8x10* J J traveling at one-half the speed of light?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





