
Concept explainers
(a)
Interpretation: Whether the following pair of formulas represents structural isomer or the same molecule should be determined:
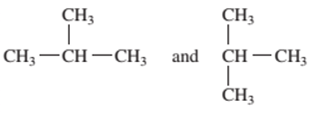
Concept Introduction:
The compounds that differ in structural formula but have same molecular formula are said to be the structural isomers.
(b)
Interpretation: Whether the following pair of formulas represent structural isomer or the same molecule should be determined:
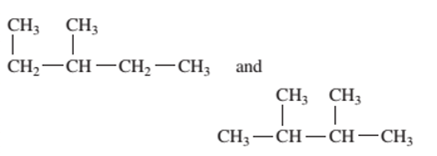
Concept Introduction:
The compounds that differ in structural formula but have same molecular formula are said to be the structural isomers.
(c)
Interpretation: Whether given pair of formulas represent structural isomer or the same molecule should be determined:

Concept Introduction:
The compounds that differ in structural formula but have same molecular formula are said to be the structural isomers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
- Draw three constitutional isomers (using line drawings) of each of the following compounds or formulasarrow_forwardidentify the following pair of organic compounds with the specific type of isomerism that they exhibit.arrow_forwardDo the following two structures represent isomers? Yes or no?arrow_forward
- How many chiral carbon atoms are present in the following compound? H3C- H3Carrow_forwardIndicate whether each of the following pairs of compounds is identical or are structural isomers of each other?arrow_forwardGive the name of each the indicated functional groups in the molecule below.arrow_forward
- How many functional groups are present in the following compound? OCH, CoO CH,arrow_forwardIdentify the functional group of the following compounds:arrow_forwardFor each of the following, state whether the two structural formulae show represent constitutional isomers or same compounds or different compounds.arrow_forward
- For each of the following pairs, indicate whether they are: the same compound different compounds that are constitutional isomers different compounds that are not isomeric.arrow_forwardWhat is the differences in physical and chemical properties of the following organic compounds? Please fill the blank C4H10 C15H32 C24H50 Reactivity > > Melting point > > Boiling point > > State of matter Molecular weight > >arrow_forwardDistinguish among the three types of isomers: structural, geometric, and enantiomerarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


