
Concept explainers
Low-molecular-weight dicarboxylic acids normally exhibit two different pKa values. Ionization of the first carboxyl group is easier than the second. This effect diminishes with molecular size, and for adipic acid and longer chain dicarboxylic acids, the two acid ionization constants differ by about one pK unit.
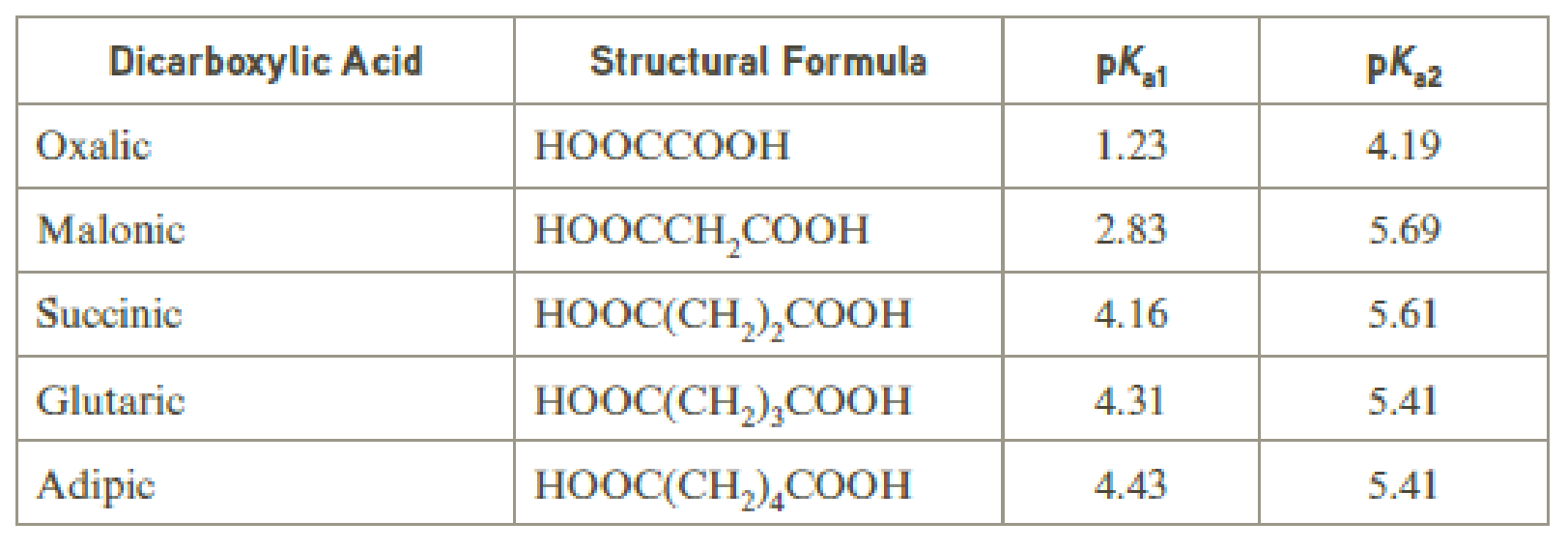
Why do the two pKa values differ more for the shorter chain dicarboxylic acids than for the longer chain dicarboxylic acids?
Trending nowThis is a popular solution!

Chapter 17 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Organic Chemistry
Chemistry: A Molecular Approach (4th Edition)
Organic Chemistry - Standalone book
EBK INTRODUCTION TO CHEMISTRY
- The pH of the given solution of lactic acid and lactate is 5.24. Calculate the pKa of lactic acid, when the concentration of lactic acid and lactate are 0.024 M and 0.087 M respectively.arrow_forwardThe following is list of weak acids and their pką values weak acid pKa benzoic acid 4.20 hydrogen citrate 6.40 hydrocyanic acid 9.31 hydrogen phosphate 12.38 Which would be the best buffer to use at pH 7 ? For benzoic acid, a 50:50 mixture of benzoic acid and its conjugate base, the benzoate ion, would have pH = The buffering capacity of a benzoic acid:benzoate buffer would be at pH = 7 A. 4.20 B. 6.40 C. 9.31 D. 12.38 E. benzoic acid F. hydrogen citrate G. hydrocyanic acid H. hydrogen phosphate I. good J. badarrow_forwardCalculate the pH of a 0.2 M solution of an amine that has a pKa of 9.5arrow_forward
- Which of the following acid/ conjugate base pairs would function best as a buffer at physiological pH? Lactic acid / Lactate ion; pka = 3.86 Carbonic acid / Bicarbonate ion; pka = 6.37 Bicarbonate ion / Carbonate ion; pKa = 10.25 Dihydrogen phosphate / Monohydrogen phosphate ion; pKa = 6.86 Acetic acid / Acetate ion; pka = 4.76arrow_forwardThe base ephedrine has a pKa value of 9.6. Calculate the theoretical end point pH when a 0.1 M solution of ephedrine is titrated with 0.1 M HCl.arrow_forwardCompare the pH of propionic acid and nitric acid when they are dissolved in water at a concentration of 20 mM. This should include writing a balanced equation describing the acid-base reactions involved, calculation of the pKa for both, calculation of pH, and calculation of the percent dissociation of both.arrow_forward
- Show the reaction and equilibrium expression for the basic dissociation of trimethylamine. Given that the Ka = 1.58 x 10-10 for trimethyl ammonium ion, use this to calculate the equilibrium constant for the basic dissociation of trimethyl amine. If the concentration of trimethylamine is 0.10 M, what is the pH of the solution?arrow_forwardDefine pKa and provide at least one method for determining its value.arrow_forwardWhat is the pH of an aqueous solution containing 0.50 mol L–1 ethanoic acid, 0.30 mol L–1 sodium ethanoate, and 0.10 mol L–1 HCl? [The pKa of ethanoic acid is 4.75.]arrow_forward
- An analytical chemist is titrating 123.4 mL of a 0.2500M solution of propylamine (C₂H-NH₂) with a 0.4100M solution of HNO3. The p K, of propylamine is 3.46. Calculate the pH of the base solution after the chemist has added 15.4 mL of the HNO3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = 0 X S ?arrow_forwardWhat is the pKa of the conjugate acid of the reaction?arrow_forwardAcid-Base Equilibria Many factors contribute to the acidity of organic compounds. Electronegativity, resonance, induction, hybridization, aromaticity, and atomic size, all play a role. In the following comparisons, you are asked to identify the factor(s) that would be most important to analyze when predicting relative acidity, and then to predict the trend in acidity and pKa values. For each of the following pairs of compounds answer the following two multiple-choice questions. 1. What factor(s) are the most important to consider when predicting the relative acidity of the two compounds? a. Electronegativity of the atom possessing the hydrogen. b. Resonance stabilization of the anionic conjugate base. c. Inductive stabilization of the anionic conjugate base. d. Hybridization of the atom possessing the hydrogen. e. The atomic size of the atom possessing the hydrogen.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

