
Concept explainers
(a)
Interpretation:
The enol form should be drawn for given compound.
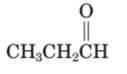
Concept Introduction:
A carbonyl compound that has a -hydrogen on a a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
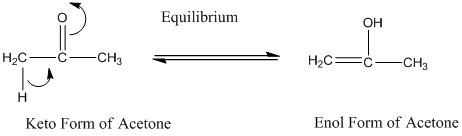
a- hydrogen is the hydrogen which attached to a a-carbon for a carbonyl compound. a-carbon is carbon adjacent to the carbonyl carbon. If a compound doesn’t have any a hydrogen is does not undergo Keto-Enol tautomerism.
(b)
Interpretation:
The enol form should be drawn for given compound.
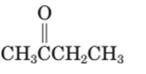
Concept Introduction:
A carbonyl compound that has a -hydrogen on a a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
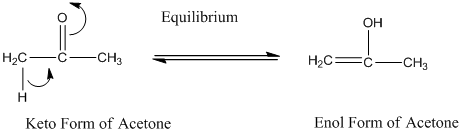
a hydrogen is the hydrogen which attached to a a-carbon for a carbonyl compound. a-carbon is carbon adjacent to the carbonyl carbon. If a compound doesn’t have any a -hydrogen is does not undergo Keto-Enol tautomerism.
(c)
Interpretation:
The enol form should be drawn for given compound.
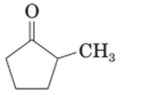 .
.
Concept Introduction:
A carbonyl compound that has a hydrogen on a a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
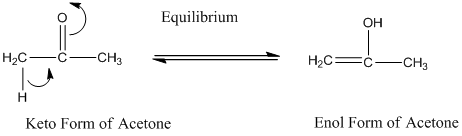
a hydrogen is the hydrogen which attached to a a-carbon for a carbonyl compound. a-carbon is carbon adjacent to the carbonyl carbon. If a compound don’t have any a hydrogen is does not undergo Keto-Enol tautomerism.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forward17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward17-29 Why can’t two molecules of acetone form a hydrogen bond with each other?arrow_forward
- 17-34 Explain why liquid aldehydes are often stored under an atmosphere of nitrogen rather than in air.arrow_forward17-79 Write an equation for each conversion. (a) 1-Pentanol to pentanal (b) 1-Pentanol to pentanoic acid (c) 2-Pentanol to 2-pentanone (d) 2-Propanol to acetone (e) Cyclohexanol to cyclohexanonearrow_forward17-33 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Pentanal and 2-pentanone (b) 2-Pentanone and 2-pentanolarrow_forward
- Draw an elimination reaction of 4-methylcyclohexanol using phosphoric acid including reaction conditions and minor products.arrow_forwardDraw the organic products formed when attached allylic alcohol A is treated with following reagent. HCrO4−–Amberlyst A-26 resinarrow_forwardHow to name an acyclic ketone using IUPAC rules ?arrow_forward
- reaction of ketones with phosphorus halidesarrow_forwardName the following ketones and aldehydes. When possible, give both a common name and an IUPAC name. CH3(CH2)5CHOarrow_forwardDraw the products formed when phenol (C6H5OH) is treated with following set of reagents. [1] CH3CH2Cl, AlCl3; [2] Br2, hνarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
