
(a)
Interpretation:
Highlight the aldehyde or ketone group in given compounds.
Concept Introduction:
Both
Here, r is alkyl group.
The general formula of ketone group is as follows:
Here, r and
Answer to Problem 17.64P
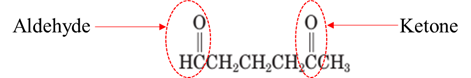
Explanation of Solution
Both aldehydes and ketones contain
(b)
Interpretation:
Highlight the aldehyde or ketone group in the given compound.
Concept Introduction:
Both aldehydes and ketones contain
Here, r is alkyl group.
The general formula of ketone group is as follows:
Here, r and
Answer to Problem 17.64P
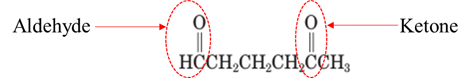
Explanation of Solution
Both aldehydes and ketones contain a
(c)
Interpretation:
Highlight the aldehyde or ketone group in the given compound.
Concept Introduction:
Both aldehydes and ketones contain
Here, r is alkyl group.
The general formula of ketone group is as follows:
Here, r and
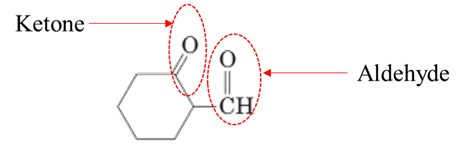
Answer to Problem 17.64P
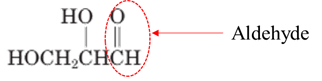
Explanation of Solution
Both aldehydes and ketones contain a

(d)
Interpretation:
Highlight the aldehyde or ketone group in the given compound.
Concept Introduction:
Both aldehydes and ketones contain
Here, r is alkyl group.
The general formula of ketone group is as follows:
Here, r and
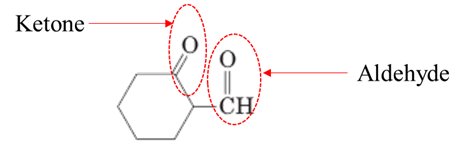
Answer to Problem 17.64P
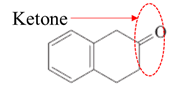
Explanation of Solution
Both aldehydes and ketones contain a
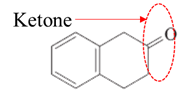
(e)
Interpretation:
Highlight the aldehyde or ketone group in the given compound.
Concept Introduction:
Both aldehydes and ketones contain
Here, r is alkyl group.
The general formula of ketone group is as follows:
Here, r and
Answer to Problem 17.64P
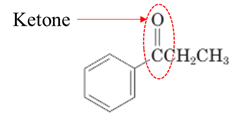
Explanation of Solution
Both aldehydes and ketones contain a
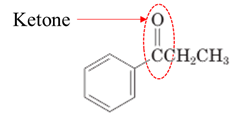
(f)
Interpretation:
Highlight the aldehyde or ketone group in the given compound.
Concept Introduction:
Both aldehydes and ketones contain
Here, r is alkyl group.
The general formula of ketone group is as follows:
Here, r and
Answer to Problem 17.64P
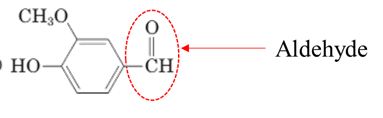
Explanation of Solution
Both aldehydes and ketones contain a
Want to see more full solutions like this?
Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-13 Which compounds contain carbonyl groups?arrow_forwardHow many carbon atoms are present in a molecule of each of the compounds in Problem 16-99? a. Methyl succinate b. Sodium succinate c. Methyl butanoate d. Methyl 2-methylbutanoatearrow_forward16-46 Arrange these three compounds in order of decreas ing ability to form intermolecular hydrogen bonds: CH3OH, CH3SH, and (CH3)2NH.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


