
Package: Organic Chemistry with Connect 2-year Access Card
5th Edition
ISBN: 9781259729959
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.62P
Answer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.
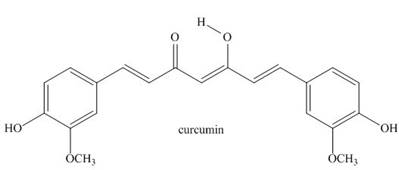
a. In Chapter 11 we learned that most enols, compounds that contain a hydroxy group bonded to a
b. Explain why the enol
c. Why is curcumin colored?
d. Explain why curcumin is an antioxidant
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Answer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.a.In Chapter 11, we learned that most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols.
b.Explain why the enol O—H proton is more acidic than an alcohol O—H proton.
c. Why is curcumin colored?
d.Explain why curcumin is an antioxidant.
Answer the following question about curcumin, a yellow pigmentisolated from turmeric, a tropical perennial in the ginger family and aprincipal ingredient in curry powder.
Most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols.
5. Describe an efficient synthesis of 1-methyl-1-cyclohexanol from cyclohexanol. Show the structure of the reagents, reactants and products from each step.
Cyclohexanol -------> -----> 1-methyl-1-cyclohexanol
Chapter 17 Solutions
Package: Organic Chemistry with Connect 2-year Access Card
Ch. 17 - Prob. 17.1PCh. 17 - Problem 17.2 What orbitals are used to form the...Ch. 17 - Problem-17.3. Give the IUPAC name for each...Ch. 17 - Prob. 17.4PCh. 17 - Problem-17.5 What is the structure of propofol,...Ch. 17 - Problem 17.6 What is the structure of a compound...Ch. 17 - Problem 17.7 How many NMR signals does each...Ch. 17 - Prob. 17.8PCh. 17 - Prob. 17.9PCh. 17 - Prob. 17.10P
Ch. 17 - Prob. 17.11PCh. 17 - Prob. 17.12PCh. 17 - Prob. 17.13PCh. 17 - Problem 17.14 Januvia, the trade name for...Ch. 17 - Prob. 17.15PCh. 17 - Problem 17.16 Rank the following compounds in...Ch. 17 - Problem 17.17 Draw the seven resonance structures...Ch. 17 - Prob. 17.18PCh. 17 - Prob. 17.19PCh. 17 - Prob. 17.20PCh. 17 - Prob. 17.21PCh. 17 - Problem 17.22 How many NMR signals does ...Ch. 17 - 17.23 Name each compound and state how many lines...Ch. 17 - Prob. 17.24PCh. 17 - Prob. 17.25PCh. 17 - Prob. 17.26PCh. 17 - 17.27 Give the IUPAC name for each compounds.
a....Ch. 17 - 17.28 Draw a structure corresponding to each...Ch. 17 - 17.29 a. Draw the 14 constitutional isomers of...Ch. 17 - Prob. 17.30PCh. 17 - Prob. 17.31PCh. 17 - Prob. 17.32PCh. 17 - 17.33 Label each compound as aromatic,...Ch. 17 - Prob. 17.34PCh. 17 - 17.35 Pentalene, azulene, and heptalene are...Ch. 17 - 17.36 The purine heterocycle occurs commonly in...Ch. 17 - Prob. 17.37PCh. 17 - 17.38
How many electrons does C contain?
How...Ch. 17 - Prob. 17.39PCh. 17 - 17.40 Explain the observed rate of reactivity of...Ch. 17 - 17.41 Draw a stepwise mechanism for the following...Ch. 17 - Prob. 17.42PCh. 17 - 17.43 Draw additional resonance structures for...Ch. 17 - Prob. 17.44PCh. 17 - Prob. 17.45PCh. 17 - 17.46 Which compound in each pair is the stronger...Ch. 17 - 17.47 Treatment of indene with forms its...Ch. 17 - Prob. 17.48PCh. 17 - 17.49 Draw the conjugate bases of pyrrole and...Ch. 17 - 17.50 a. Explain why protonation of pyrrole occurs...Ch. 17 - Prob. 17.51PCh. 17 - Prob. 17.52PCh. 17 - 17.53 How many signals does each compound...Ch. 17 - 17.54 Which of the diethylbenzene isomers (ortho,...Ch. 17 - 17.55 Propose a structure consistent with each...Ch. 17 - 17.56 Propose a structure consistent with each...Ch. 17 - 17.57 Thymol (molecular formula ) is the major...Ch. 17 - 17.58 You have a sample of a compound of molecular...Ch. 17 - 17.59 Explain why tetrahydrofuran has a higher...Ch. 17 - 17.60 Rizatriptan (trade name Maxalt) is a...Ch. 17 - 17.61 Zolpidem (trade name Ambien) promotes the...Ch. 17 - 17.62 Answer the following questions about...Ch. 17 - 17.63 Stanozolol is an anabolic steroid that...Ch. 17 - Prob. 17.64PCh. 17 - 17.65 Use the observed data to decide whether C...Ch. 17 - Prob. 17.66PCh. 17 - Prob. 17.67PCh. 17 - Prob. 17.68PCh. 17 - 17.69 Although benzene itself absorbs at in its ...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What explains why many aldehydes and ketones can undergo self- condensation reactions in basic conditions? A. The alpha carbon can lose a proton and act like a nucleophile and the carbonyl carbon a an electrophile B. The alpha carbon can gain a proton and act like an electrophile and the carbonyl carbon is a nucleophile C. The oxygen of the carbonyl group can attack the carbon of the carbonyl group D. Only esters can undergo self-condensation reactionsarrow_forwardAcetals are formed from the reaction of two alcohols with a carbonyl under acidic conditions. Acetal formation is faster with 1,2-ethanediol than with two methanol molecules. Choose the factor that explains the difference in reaction rates. A) The reaction with 1,2-ethanediol has a lower AH (enthalpy) of reaction. B) The reaction with 1,2-ethanediol has a higher AH (enthalpy) of reaction. C) The reaction with 1,2-ethanediol has a more favorable entropy of reaction.arrow_forwardMaltose is a carbohydrate present in malt, the liquid obtained from barley and other grains. Although maltose has numerous functional groups, its reactions are explained by the same principles we have already encountered.a. Label the acetal and hemiacetal carbons.b. What products are formed when maltose is treated with each of these reagents: [1] H3O+; [2] CH3OH and HCl; [3] excess NaH, then excess CH3I?c. Draw the products formed when the compound formed in Reaction [3] of part (b) is treated with aqueous acid.The reactions in parts (b) and (c) are used to determine structural features of carbohydrates like maltose.arrow_forward
- 1. What reagents would cause the following conversions? OH H. H. OH H. HO, 2. What is the mechanism for acetal/ketal formation? OH HO TSOH TSOHarrow_forwardDraw the enol form of the following ketonearrow_forwardDraw out each compound to clearly show what groups are bonded to the carbonyl carbon. Label each compound as a ketone or aldehyde. a. CH 3CH 2CHO b. CH 3CH 2COCH 3 c. (CH 3) 3CCOCH 3 d. (CH 3CH 2) 2CHCHOarrow_forward
- Draw two enol forms of the following diketone.arrow_forward2: Draw the structure of the starting materíals and reagents (A, B, C) needed to prepare the given product. 1. C но Mg, ether A 2. H,0arrow_forwardDraw the tautomer of this enol. Include all lone pairs. Ignore inorganic byproducts. :OH: H3O+ Draw Tautomerarrow_forward
- What two functional groups react to form the following? a. A hemiacetal b. An acetal c. A ketal d. A hemiketalarrow_forwardCompounds that contain both a hydroxyl group (OH) and a carboxyl group (COOH) can undergo an intramolecular esterifi cation reaction. What product is formed when each hydroxy acid undergoes an intramolecular reaction? a. HOCH 2CH 2CH 2CH 2CO 2H b. HOCH 2CH 2CH 2CO 2Harrow_forwardDraw the most stable product formed in each of the reactions shown. Reaction (a) 2 NaOEt, EtOH A Draw the product of reaction (a).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License