
(a)
Interpretation:
Structure of amine or quaternary ammonium salt that is obtained when dimethylamine and propyl bromide is reacted in presence of strong base has to be drawn.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering
(a)
Answer to Problem 17.70EP
The structure of product obtained is,
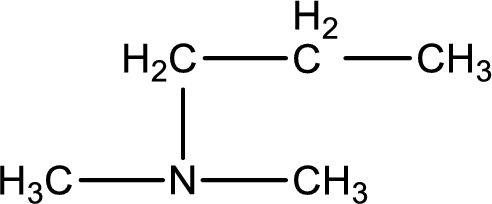
Explanation of Solution
Given reactants are dimethylamine and propyl bromide. In this, the amine given is a secondary amine. When secondary amine is treated with an alkyl halide, the product obtained is a tertiary amine. The complete reaction and the structure of the product formed is given as,
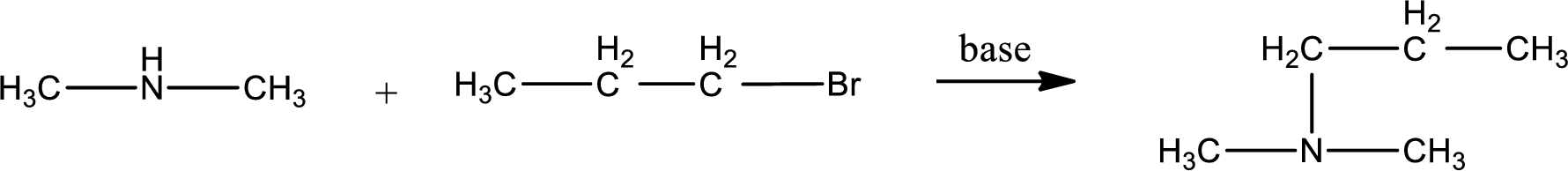
Structure of the product formed is drawn.
(b)
Interpretation:
Structure of amine or quaternary ammonium salt that is obtained when diethylmethylamine and isopropyl chloride is reacted in presence of strong base has to be drawn.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering amines, the alkylating agent that is used is alkyl halides. Alkylation is done under basic conditions. The general equations for amines alkylation process is,
(b)
Answer to Problem 17.70EP
The structure of product obtained is,
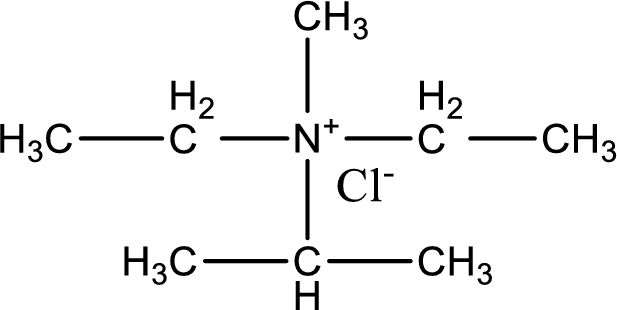
Explanation of Solution
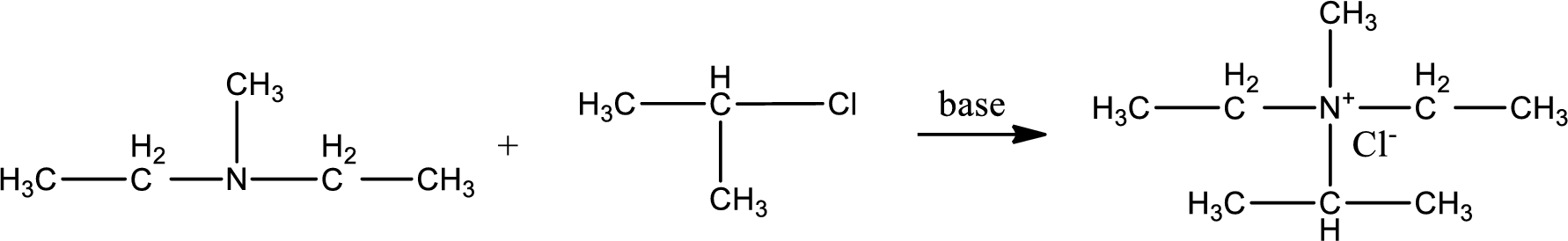
Given reactants are diethylmethylamine and isopropyl chloride. In this, the amine given is a tertiary amine. When tertiary amine is treated with an alkyl halide, the product obtained is a quaternary ammonium salt. The complete reaction and the structure of the product formed is given as,
Structure of the product formed is drawn.
(c)
Interpretation:
Structure of amine or quaternary ammonium salt that is obtained when methylpropylamine and ethyl chloride is reacted in presence of strong base has to be drawn.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering amines, the alkylating agent that is used is alkyl halides. Alkylation is done under basic conditions. The general equations for amines alkylation process is,
(c)
Answer to Problem 17.70EP
The structure of product obtained is,
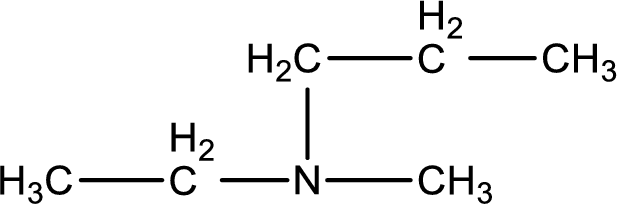
Explanation of Solution
Given reactants are methylpropylamine and ethyl chloride. In this, the amine given is a secondary amine. When secondary amine is treated with an alkyl halide, the product obtained is a tertiary amine. The complete reaction and the structure of the product formed is given as,
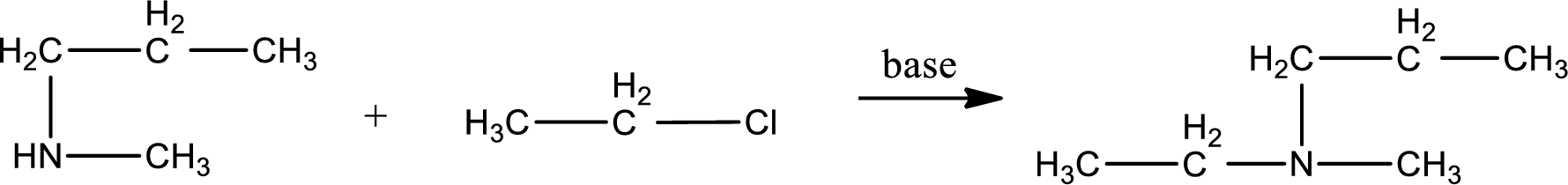
Structure of the product formed is drawn.
(d)
Interpretation:
Structure of amine or quaternary ammonium salt that is obtained when tripropylamine and propyl chloride is reacted in presence of strong base has to be drawn.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering amines, the alkylating agent that is used is alkyl halides. Alkylation is done under basic conditions. The general equations for amines alkylation process is,
(d)
Answer to Problem 17.70EP
The structure of product obtained is,
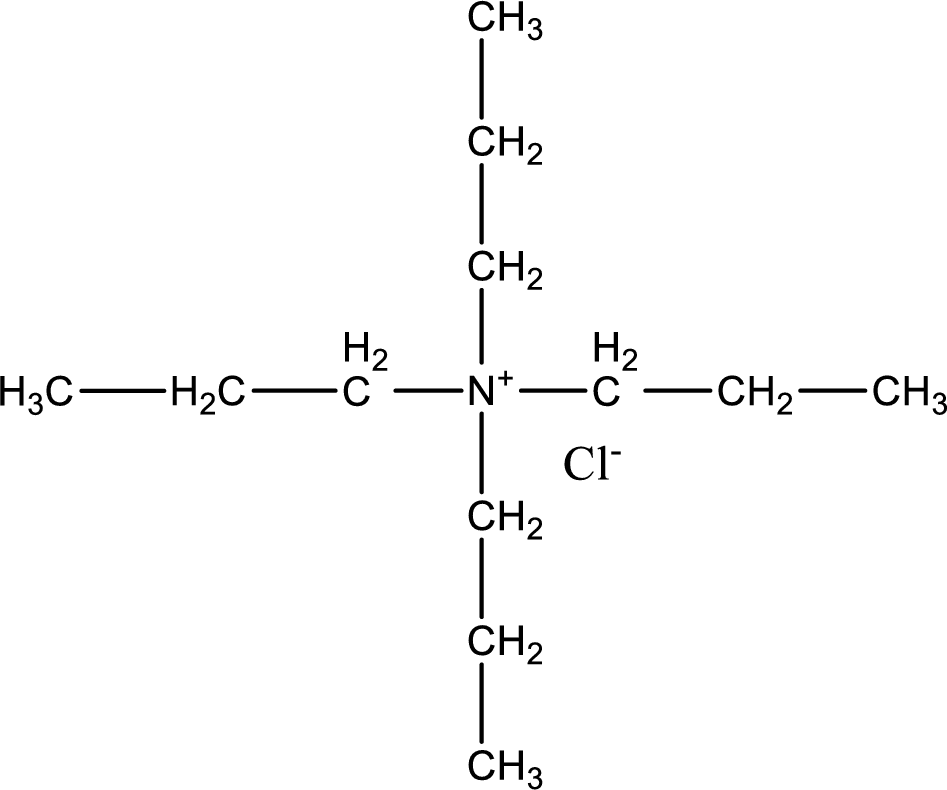
Explanation of Solution
Given reactants are tripropylamine and propyl chloride. In this, the amine given is a tertiary amine. When tertiary amine is treated with an alkyl halide, the product obtained is a quaternary ammonium salt. The complete reaction and the structure of the product formed is given as,
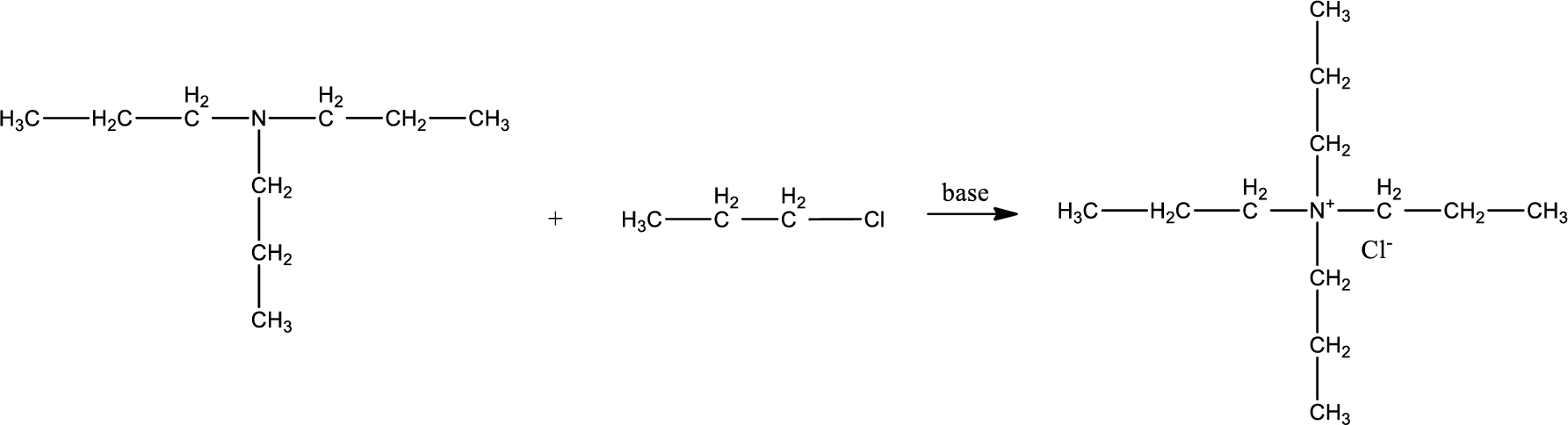
Structure of the product formed is drawn.
Want to see more full solutions like this?
Chapter 17 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Salicylic acid (o-hydroxybenzoic acid) is used as starting material to prepare aspirin. Draw the structure of salicylic acid.arrow_forwardDetermine the weight/volume of the chemicals needed to prepare the following solutions: a) 100 ml of 0.9% (w/v) saline (NaCl) b) 30 ml of 50% glycerol (v/v) c) Electrophoresis requires TAE, which is a specific mixture of Tris base, acetic acid, and EDTA. TAE is normally made as a 50X concentrated stock. Provide a recipe to make 40 ml of 50X TAE. The recipe for one liter of 50X TAE is as follows: 242g Tris base, 57.1 ml glacial acetic acid, 100 ml 0.5 M EDTAarrow_forwardScott test is one of the methods used for screening for Cocaine in Forensic Chemistry. The multi step method consists of a) reacting the sample drug with cobaltous thiocyanate (Co(SCN)2) b) dissolving of the formed precipitate by adding HCl and c) extraction of coordination compound with Chloroform d) measurement of absorbance of extracted coordination compound at 627 nm What is the type of chemical equilibria involved in first step of the method? a.Solubility Product equilibria b.Bronsted Lowry acid – Bronsted Lowry base equilibria c.Complex ion formation d.Coordination compound formationarrow_forward
- Consider these compounds: A. PbBr, B. MnS C. Ag,CO3 D. AIPO, Complete the following statements by entering the letter(s) corresponding to the correct compound(s). (If more than one compound fits the description, include all the relevant compounds by writing your answer as a string of characters without punctuation, e.g, ABC.) Without doing any calculations it is possible to determine that magnesium fluoride is more soluble than and magnesium fluoride is less soluble than| It is not possible to determine whether magnesium fluoride is more or less soluble than by simply comparing Kgp values.arrow_forwardThe ionization of p-nitrophenol is shown below (pKa = 7.0): a. Identify the weak acid and conjugate base. b. At pH 7, what are the relative concentrations of ionized and un-ionized p-nitrophenol? c. If enough concentrated hydrochloric acid is added to a solution of p-nitrophenol to lower the pH from 7 to 5, what will happen to the relative concentrations of the ionized and un-ionized forms? d. Ionized p-nitrophenol has a yellow color, while the un-ionized form is colorless. The yellow color can be measured using a spectrophotometer at 400nm. In order to determine the total amount of p-nitrophenol in a solution, would you perform the spectrophotometer reading at an acidic or basic pH? Clearly explain why? e. A solution of p-nitrophenol at pH 7.95 was found to have an A400 of 0.255 . What is the total concentration (in µM) of p-nitrophenol (ionized plus un-ionized) in the solution? The molar extinction coefficient of p-nitrophenol is 18,500 M-1cm-1 and the pKa is 7.arrow_forwardWrite the chemical formula of the conjugate base of boric acid.arrow_forward
- Give the reagent or chemical test that would differentiate the following pairs of compounds. Write chemical equations for the reactions. List of Chemical Tests (choose here)I. Halogenation: Bromination with and without lightII. Oxidation: Reaction with Baeyer’s reagent III. Reaction with Ammoniacal Silver Nitrate (AgNO3) or simply Tollen’s reagent a. benzene and ethylbenzeneb. 1-butyne and 2-butynec. 2-methylpentane and 2-methyl-2-pentened. toluene and 1-methylcyclohexenearrow_forwardDraw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardIdentify the conjugate acid-base pairs in the following reactions: HNO2(aq) + H2O(l) → NO2 – (aq) + H3O+(aq) _______ ______ _________ ________ CH3NH2 + H2O(l) → CH3NH3+ + OH – _______ ________ ________ _________arrow_forward
- Indicate what you would do and the observations you would expect for each case. Be explicit in your explanations . Use simple chemical tests ( not including melting point, boiling point, spectroscopy, etc.) whenever possible and more elaborate tests for other cases. Describe how to distinguish between each of the following: A. 1-phenylpropyne and 3-phenylpropyne B. phenol and p-hydroxybenzyl alcoholarrow_forwardThe reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardConsider a buffer solution of acetate. The volume is 500 ml, the concentration is 200 mM, and the pH is 5.0. a. How many total moles of acetate plus acetic acid are present in the solution? Express answer as x.y with one digit before and one after the decimal place. b. What is the ratio of acetate ions (Ac-) to acetic acid ions (HAc) in the buffer solution (pH 5.0) if the pka is 4.76? Express the answer as x.yz with one digit before and two after the decimal place. c. How many moles of acetate are present in the solution? Express your answer to one decimal place. d. How many moles of acetic acid are present in the solution? Express your answer to one decimal place.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





