
Concept explainers
Practice Problem 17.1
Give an IUPAC systematic name for each of the following:
(a)
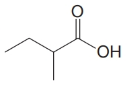
(b)
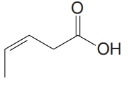
(c)
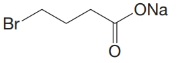
(d)
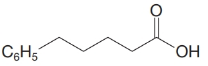
(e)
Interpretation:
The IUPAC name for each given carboxylic compound is to be identified.
Concept introduction:
The carboxylic group
Answer to Problem 1PP
Solution:
Explanation of Solution
a)

Start numbering the chain beginning from the carbon of the carboxylic group.
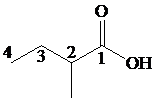
This chain contains four carbons and so the base name is butanoic acid. In the position-2, it has a methyl group attached. Therefore, the compound is
b)
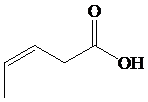
The numbering of the chain begins from the carbon of the carboxylic group.
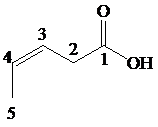
This chain contains five carbons. So, the compound belongs to pentanoic acid. Here, in position-3, it has a double bond. Thus, by considering this double bond and its stereochemistry, the compound identified is
c)

The numbering of the chain begins from the carbon of the carboxylic group.
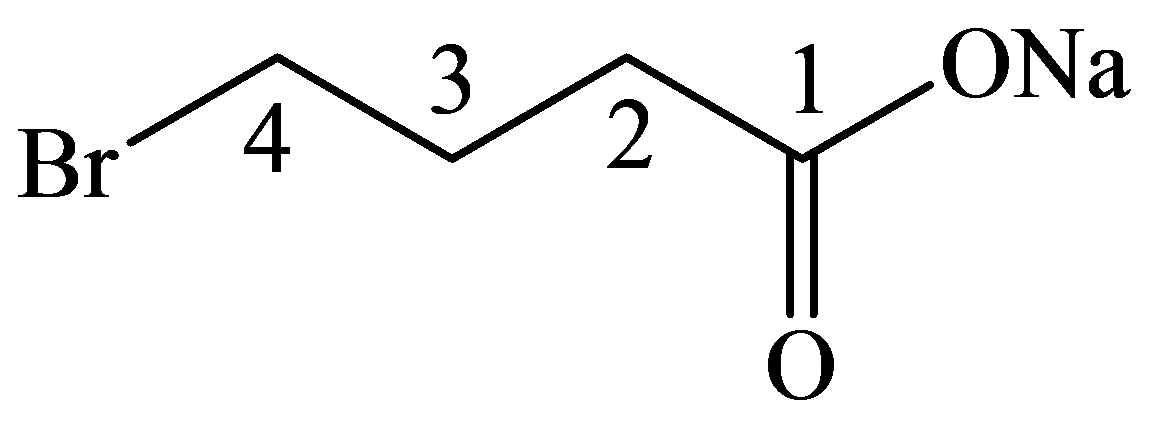
This chain contains four carbons, and so, the name of the compound must be butane. Here, the hydrogen of the carboxylic group is replaced by sodium ion
d)
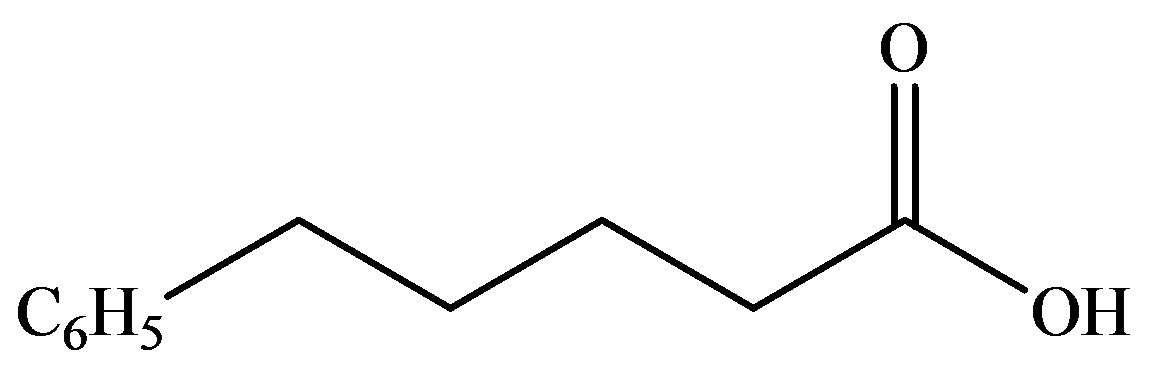
The numbering of the chain begins from the carbon of the carboxylic group.
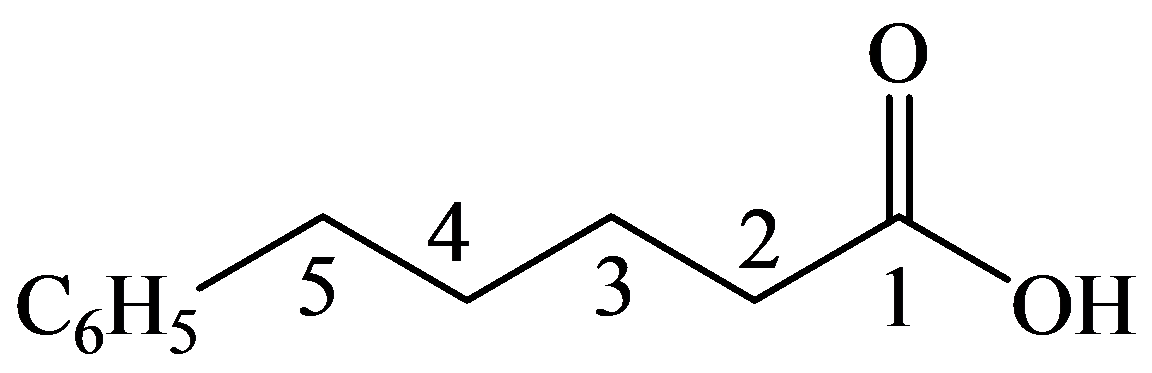
This chain contains five carbons, and so, it is pentanoic acid. It has a phenyl compound at position-5. Thus, the name of the compound is
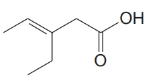
e)

The numbering of the chain begins from the carbon of the carboxylic group.
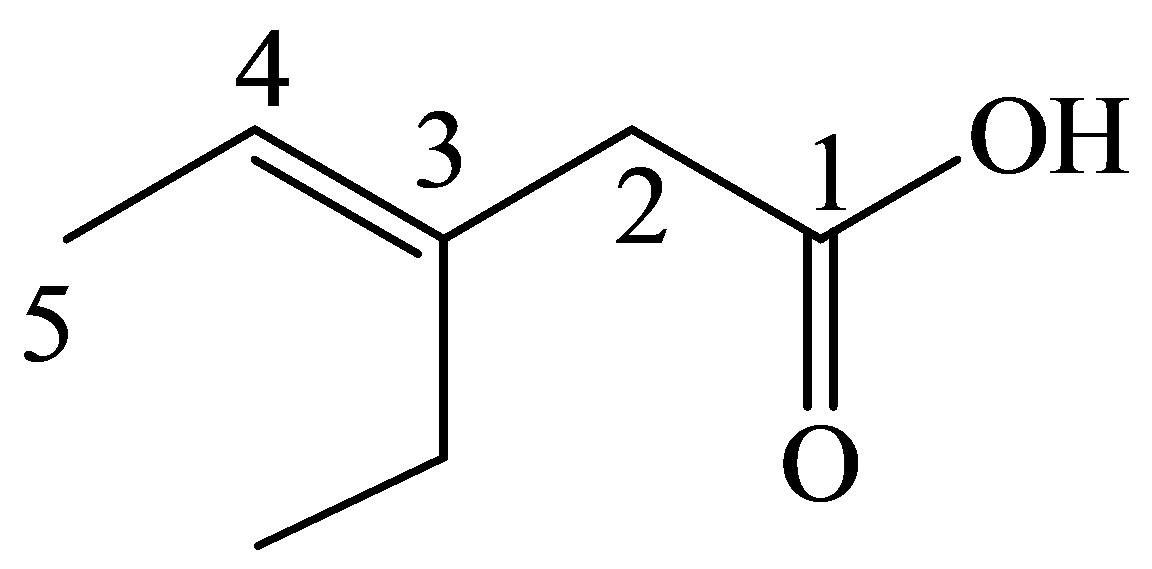
This chain contains five carbons. So, the name is pentanoic acid. There is a double bond and an ethyl group in position-3. Therefore, by considering the above conditions and the stereochemistry involved, the name of the compound is
Want to see more full solutions like this?
Chapter 17 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Additional Science Textbook Solutions
Chemistry: A Molecular Approach (4th Edition)
Chemistry
Chemistry & Chemical Reactivity
Chemistry (7th Edition)
Principles of Chemistry: A Molecular Approach (3rd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
- Provide IUPAC names for the following compounds.(a) (CH3)2CHCH2CH3 (b) CH3¬C(CH3)2¬CH3arrow_forward11:43 Q1. (a) (c) (d) (b) Two stereoisomers of but-2-ene are formed when 2-bromobutane reacts with ethanolic potassium hydroxide. (i) Explain what is meant by the term stereoisomers. Library Name and outline a mechanism for the reaction of 2-bromo-2-methylpropane with ethanolic potassium hydroxide to form the alkene 2-methylpropene, (CH3)2C=CH₂ Name of mechanism Mechanism (ii) Draw the structures and give the names of the two stereoisomers of but-2-ene. Stereoisomer 1 Name (iii) Name this type of stereoisomerism. Select Name Stereoisomer 2 When 2-bromo-2-methylpropane reacts with aqueous potassium hydroxide, 2-methylpropan-2-ol is formed as shown by the following equation. CH3 H₂C-C-CH3 + KOH Br Page 2 of 14 CH3 H3C-C-CH3 + KBr ОН State the role of the hydroxide ions in this reaction. Write an equation for the reaction that occurs when CH3CH₂CH₂CH₂Br reacts with an excess of ammonia. Name the organic product of this reaction. Equation Name of product 9,284 Photos, 1,166 Videos For You…arrow_forwardProvide the reagents and solvents (where appropriate) needed to bring about the following transformations. (a) CI (b)arrow_forward
- 5.34 Select the compound in each of the following pairs that will be converted to the corresponding alkyl bromide more rapidly on being treated with hydrogen bromide. Explain the reason for your choice. (a) 1-Butanol or 2-butanol (b) 2-Methyl-1-butanol or 2-butanol (c) 2-Methyl-2-butanol or 2-butanol (d) 2-Methylbutane or 2-butanol dovolonentanol or cyclohexanolarrow_forwardGive the series of reactions below, identify and give the iupac name for the following compounds, in short identify and name A,B,Carrow_forwardD.32 Write the IUPAC name for each of the following compounds, including stereochemical designations. (a) (b) (c) HO NH2 OH OCH3arrow_forward
- Rank the compounds in each group according to their reactivity towardelectrophilic substitution.(a) Chlorobenzene, o-dichlorobenzene, benzene(b) p-Bromonitrobenzene , nitrobenzene, phenol(c) Fluorobenzene, benzaldehyde, a-xylene(d) Benzonitrile, p-methylbenzonitr ile,p-methoxybenzonitrilearrow_forwardWhich of the following could be used as a solvent in the deprotonation reaction of terminal alkynes? A CH;OH (I) B CH;COOH (I) NH3 (1) D) H20 ()arrow_forward4.33 Select the compound in each of the following pairs that will be converted to the corresponding alkyl bromide more rapidly on being treated with hydrogen bromide. Explain the reason for your choice. (a) 1-Butanol or 2-Butanol (b) 2-Methyl-1-butanol or 2-butanol (c) 2-Methyl-2-butanol or 2-butanol (d) 2-Methylbutane or 2-butanol (e) 1-Methylcyclopentanol or cyclohexanol Draw the energy diagrams of an SN1 reaction and an SN2 reaction. Include in your drawing anexample reaction. Identify the rate limiting step and label it as unimolecular or bimolecular.arrow_forward
- [ 16 ] which of following compound(s) is/are neither acetal nor hemiacetal ? он Но Он осH-CHз CHз ОН OCH3 (1) ( II ) ( III ) (IV ) (V) (a ) compounds III and V (b) compounds Il and II (c) compounds I and V (d ) compounds IV and V (e) compounds I and IVarrow_forwardAttenol A and pinnatoxin A are natural products isolated from marine sources. (a) Locate the acetals, hemiacetals, imines, and enamines in both compounds. (b) Draw the hydrolysis product formed when attenol A is treated with aqueous acid. Include stereochemistry at all stereogenic centers.arrow_forward5.20 Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3),CHCH2CH2CH2B1 (b) (CH3)2 CHCH2CH2 CH2OH (c) Cl3 CCH2B C12CHCHB1 (d) Cl (e) CF3CH2OH (f) ОН CH3 (g) ОН CH3 (h) Br (i) ОНarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





