
(a)
Interpretation: The equation for the reaction of acid in Example 17.3 with ammonia has to be written and the name of the
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
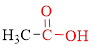
Nomenclature of carboxylic acid:
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the
alkane name with –oic acid.
Carboxylic acid reacts with ammonia gives neutral carboxylic salt.
(b)
Interpretation: The equation for the reaction of acid in Example 17.3 with ammonia has to be written and the name of the carboxylic acid salt formed should be named.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,

Nomenclature of carboxylic acid:
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the alkane name with –oic acid.
Carboxylic acid reacts with ammonia gives neutral carboxylic salt.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Write an equation for the reaction of chloroacetic acid (Ka=1.5103) with trimethylamine (Kb=5.9105) . Calculate the equilibrium constant for the reaction. If 0.10 M solutions of these two species are mixed, what will be their concentrations at equilibrium?arrow_forwardWrite the structural formulas of trichloroacetic acid and acetic acid and explain why trichloroacetic acid is the much stronger acid of the two.arrow_forwardSalicylic acid preparation and propertiesarrow_forward
- Complete the following acid-base reactions.arrow_forwardWrite the chemical equation between acetic acid and sodium hydroxide.arrow_forwardDraw the Lewis structures for chloroacetic acid and for acetic acid. Use structural arguments to explain why acetic acid is a weaker acid then chloroacetic acid. Be sure to refer to the stability of the ion that would be formed after dissociation of the acid.arrow_forward
- The purity of the aspirin sample is tested with iron (III) chloride. A purple color resulting from iron (III) chloride test indicates the aspirin sample contains some unreacted salicylic acid and the carboxylic acid group in salicylic acid gives a purple color. the structure of aspirin itself contains a phenolic -OH group. O the aspirin sample contains some unreacted salicylic acid and the phenolic -OH group in salicylic acid gives a purple color. the aspirin sample contains some unreacted salicylic acid and the ring structure in salicylic acid gives a purple color.arrow_forwardWrite equations for the reaction of acid with ammonia and name the carboxylic salt formed.arrow_forwardcarboxylic acids react with water to form sodium hydroxide True False carboxylic acids react with bases such as sodium hydroxide to form carboxylate salts and water True False carboxylic acids are strong acids that dissociate completely to form carboxylate ions in water True Falsearrow_forward
- write the esterification reaction of lauric acid with ethanolarrow_forwardWrite properties of ester (structural and physiochemical). Name one ester used in chewing gum. Explain comprehensively.arrow_forwardEster compounds often have a sweet, pleasant odor. Many characteristic fruit scents are largely due to the natural presence of one or more ester compounds. As such, artificial scents for foods are often composed of complex mixtures of various esters. The exact identity and ratio of ingredients that compose a particular scent are closely guarded secrets in the food and fragrance industry. Suppose that you are a chemist working for a company that i creating a new line of air fresheners. The company is considering three scents: apple, pear, and pineapple. The project manager has asked you to prepare the ester compounds that are largely responsible for these scents. The structural formulas for these ester compounds are shown here: Alcohols for Air Freshener Project Molar mass Density Cost, per (g/mL) Reagent (g/mol) 1.00 L methanol 32.04 0.79 $46.20 ethanol 46.07 0.79 $112.00 1-propanol 60.10 0.80 $72.70 1-butanol 74.12 0.81 $72.60 Use the structural formulas of the alcohols and…arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





