
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 18.14P
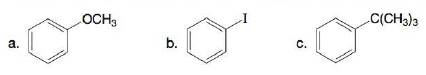
Classify each substituent as electron donating or electron withdrawing.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Click the "draw structure" button to launch the drawing utility.
Draw the product formed when the following a.f-unsaturated ketone is treated with
[1] (CH,-CH),CuLi; [2] H,O.
draw structure .
Label attached pair of compounds as keto–enol tautomers or constitutionalisomers, but not tautomers.
14) Identiff and circle any acetals, ketals, hemiacetals and hemi ketals present in the followingMolecule
Chapter 18 Solutions
Organic Chemistry-Package(Custom)
Ch. 18 - Prob. 18.1PCh. 18 - Prob. 18.2PCh. 18 - Prob. 18.3PCh. 18 - Prob. 18.4PCh. 18 - Prob. 18.5PCh. 18 - What acid chloride would be needed to prepare each...Ch. 18 - Prob. 18.7PCh. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.9PCh. 18 - Prob. 18.10P
Ch. 18 - Prob. 18.11PCh. 18 - Prob. 18.12PCh. 18 - Problem 18.14 Draw all resonance structures for...Ch. 18 - Classify each substituent as electron donating or...Ch. 18 - Prob. 18.15PCh. 18 - Label each compound as more or less reactive than...Ch. 18 - Rank the following compounds in order of...Ch. 18 - Problem 18.18 Rank the following compounds in...Ch. 18 - Prob. 18.19PCh. 18 - Problem 18.20 Draw the products of each...Ch. 18 - Prob. 18.21PCh. 18 - Prob. 18.22PCh. 18 - Devise a synthesis of each compound from the...Ch. 18 - Problem 18.24 Draw the products of each...Ch. 18 - Problem 18.25 Draw a stepwise mechanism for the...Ch. 18 - Problem 18.26 Draw the products of each...Ch. 18 - Prob. 18.27PCh. 18 - Prob. 18.28PCh. 18 - Problem 18.29 How could you use ethylbenzene to...Ch. 18 - Prob. 18.30PCh. 18 - Problem 18.31 What steps are needed to convert...Ch. 18 - Problem 18.32 Synthesize each compound from...Ch. 18 - Synthesize each compound from benzene.Ch. 18 - Prob. 18.34PCh. 18 - 18.35 What is the major product formed by an...Ch. 18 - Draw the products formed when phenol (C6H5OH) is...Ch. 18 - Prob. 18.37PCh. 18 - Draw the products of each reaction. a. e. i. b. f....Ch. 18 - What products are formed when benzene is treated...Ch. 18 - Draw the products of each reaction. a.d. b.e. c.f.Ch. 18 - You have learned two ways to make an alkyl...Ch. 18 - Prob. 18.42PCh. 18 - Prob. 18.43PCh. 18 - 18.45 Explain why each of the following reactions...Ch. 18 - Prob. 18.45PCh. 18 - 18.47 For each of the following substituted...Ch. 18 - Which benzene ring in each compound is more...Ch. 18 - 18.49 For each N-substituted benzene, predict...Ch. 18 - Explain each statement in detail using resonance...Ch. 18 - Prob. 18.50PCh. 18 - Explain why the meta product is formed in the...Ch. 18 - 18.53 Rank the aryl halides in each group in order...Ch. 18 - Prob. 18.53PCh. 18 - 18.54 Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.55PCh. 18 - 18.56 Draw a stepwise, detailed mechanism for the...Ch. 18 - Prob. 18.57PCh. 18 - 18.58 Draw a stepwise mechanism for the following...Ch. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.60PCh. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.62PCh. 18 - Prob. 18.63PCh. 18 - Synthesize each compound from benzene and any...Ch. 18 - Synthesize each compound from benzene and any...Ch. 18 - Synthesize each compound from toluene (C6H5CH3)...Ch. 18 - Prob. 18.67PCh. 18 - Use the reactions in this chapter along with those...Ch. 18 - Prob. 18.69PCh. 18 - Prob. 18.70PCh. 18 - Problem 18.69 Identify the structures of isomers A...Ch. 18 - Prob. 18.72PCh. 18 - Problem 18.71 Compound X (molecular formula ) was...Ch. 18 - Prob. 18.74PCh. 18 - The NMR spectrum of phenol () shows three...Ch. 18 - Explain the reactivity and orientation effects...Ch. 18 - Prob. 18.77PCh. 18 - Prob. 18.78PCh. 18 - Prob. 18.79PCh. 18 - Prob. 18.80P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- IUPAC namearrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] HC≡CNa; [2] H2Oarrow_forward1. Draw the alicyclic hydroxylated metabolite for the following drug H3C. 2. Draw the lactam metabolite from following drugarrow_forward
- dont provide handwritiing solution ..arrow_forwardDraw the compound shown in the hemiacetal form as a pyranose in the more stable chair form. ww HO- HO. HO- HO- HO-arrow_forwardDraw the hydrogen bonding of G-C and A-T pairs by hand. For each hydrogen bond, please point out which are hydrogen bond donors, and which are hydrogen bond acceptors.arrow_forward
- Draw the organic products formed in the following reaction.arrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] CH3C≡CLi; [2] H2Oarrow_forwardwhich compound can be classified as an Acetal OH a) b) OH (c) Y d)arrow_forward
- Draw a tautomer of the molecule pictured. NH₂arrow_forwardDraw the major products of the SN1 reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, where applicable. Ignore any inorganic byproducts. H OH HCI Q Please select a drawing or reagent from the question areaarrow_forwardUse the following wedge formula mentioned in attached diagram to answer the following question. Draw one enantiomer and two diastereomers of structure (ii).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY