
Concept explainers
Following is a retrosynthetic analysis for the synthesis of the herbicide (S)-Metolachlor from 2-ethyl-6-methylaniline, chloroacetic acid, acetone, and methanol.
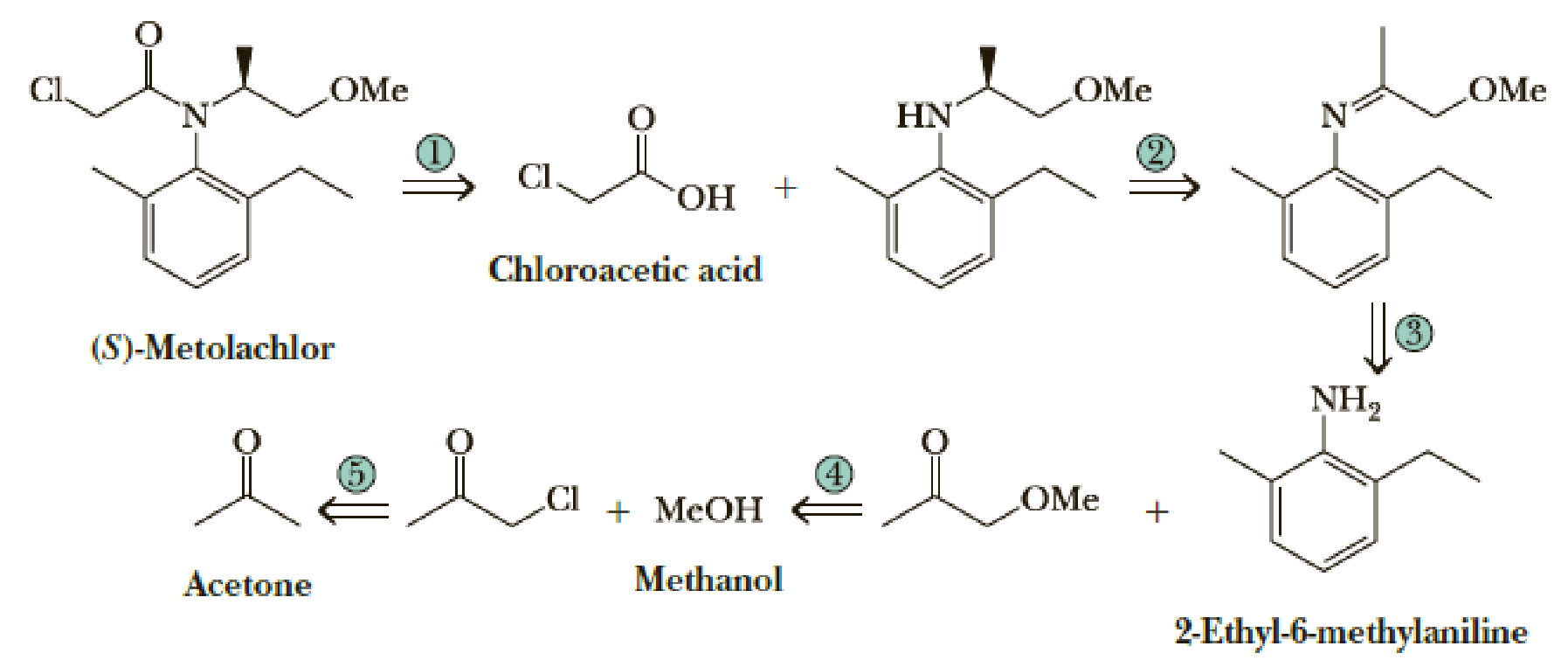
Show reagents and experimental conditions for the synthesis of Metolachlor from these four organic starting materials. Your synthesis will most likely give a racemic mixture. The chiral catalyst used by Novartis for reduction in Step 2 gives 80% enantiomeric excess of the S enantiomer.
Trending nowThis is a popular solution!

Chapter 18 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Additional Science Textbook Solutions
Essential Organic Chemistry (3rd Edition)
Chemistry: Structure and Properties
Chemistry
Organic Chemistry
Inorganic Chemistry
General, Organic, and Biological Chemistry (3rd Edition)
- Prepare the following compounds starting from benzaldehyde and the appropriate ketone. Provide reactions for preparing the ketones starting from aromatic hydrocarbon compounds.arrow_forwardSynthetize 3-phenyl-2-propenoic acid from benzaldehyde using whatever organic/inorganic reagents are needed. 3-phenyl-2-propenoic = 3-phenylacrylic acidarrow_forwardDescribe how 3-methyl-1-phenyl-3-pentanol can be prepared from benzene. You can use any inorganic reagents and solvents, and any organic reagents provided they contain no more than two carbons.arrow_forward
- Describe a laboratory procedure that would allow you to form 2-pentanone and 3-pentanone from 2-pentyne. Show a synthesis pathway in your description, including the full structural diagrams for all reactants, products, and intermediatesarrow_forwardHow would you perform an experiment to produce cyclohexanol from cyclohexene? Describe in detail the reagents and glassware you would use including approximate amounts and concentrations of reagents. What steps would be needed to purify the cyclohexanol from the other reagents used in the reaction?arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forward
- Following is a retrosynthetic analysis for an intermediate in the industrial synthesis of vitamin A. (a) Addition of one mole of HCl to isoprene gives 4-chloro-2-methyl-2-butene as the major product. Propose a mechanism for this addition and account for its regioselectivity. (b) Propose a synthesis of the vitamin A precursor from this allylic chloride and ethyl acetoacetate.arrow_forwardShow how to bring about each step in this synthesis of the herbicide propranil.arrow_forwardThe pyrolysis of acetic esters to give an alkene and acetic acid is thought to involve a planar transition state and cyclic redistribution of (4n + 2) electrons. Propose a mechanism for pyrolysis of the following ester.arrow_forward
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardAccount for the fact that treating propenoic acid (acrylic acid) with HCl gives only 3-chloropropanoic acid.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
