
(a)
Interpretation:
The way by which the given alcohol is prepared by treating an ester with a Grignard reagent has to be shown.
Concept introduction:
Alkyl or aryl magnesium halides
Reaction of Grignard reagent with a formic ester followed by hydrolysis in aqueous acid yields secondary alcohol. Tertiary alcohol can be prepared by the reaction of Grignard reagent with ester.
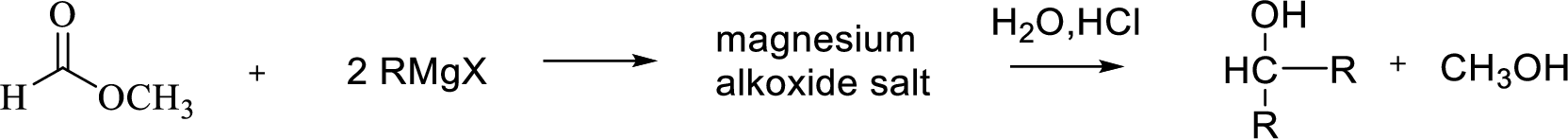
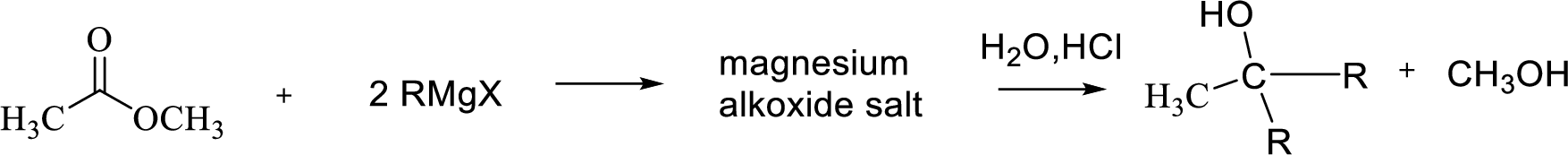
(b)
Interpretation:
The way by which the given alcohol is prepared by treating an ester with a Grignard reagent has to be shown.
Concept introduction:
Alkyl or aryl magnesium halides
Reaction of Grignard reagent with a formic ester followed by hydrolysis in aqueous acid yields secondary alcohol. Tertiary alcohol can be prepared by the reaction of Grignard reagent with ester other than formate.
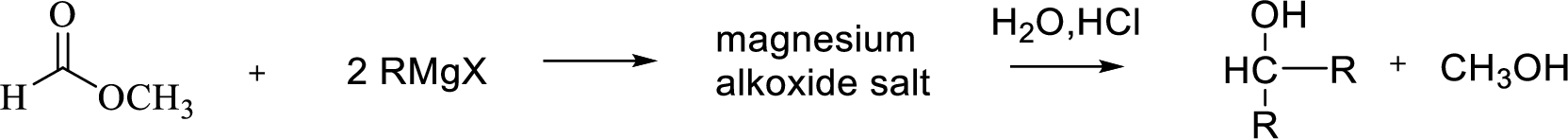
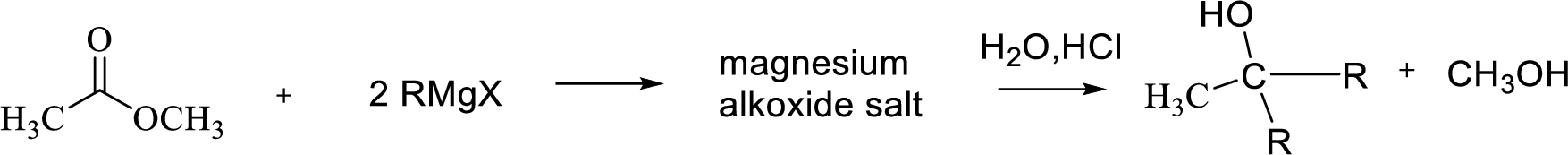
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Show how each alcohol or diol can be prepared from an alkene. (a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol (d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol (g) 1,2-Hexanediolarrow_forwardAcetals are formed from the reaction of two alcohols with a carbonyl under acidic conditions. Acetal formation is faster with 1,2-ethanediol than with two methanol molecules. Choose the factor that explains the difference in reaction rates. A) The reaction with 1,2-ethanediol has a lower AH (enthalpy) of reaction. B) The reaction with 1,2-ethanediol has a higher AH (enthalpy) of reaction. C) The reaction with 1,2-ethanediol has a more favorable entropy of reaction.arrow_forwardHow Enols React ?arrow_forward
- Why can’t 2-methyl-2-propanol be prepared by the reduction of a carbonyl compound?arrow_forwardDraw an elimination reaction of 4-methylcyclohexanol using phosphoric acid including reaction conditions and minor products.arrow_forwardDraw the products obtained by reacting a sulfonitric mixture with: 4-nitrophenol, 2,4-dinitrophenol, phenol, 4-methoxyphenol, 4-chlorophenol.arrow_forward
- Show how to convert alkenes, alkyl halides, and carbonyl compounds to alcohols.arrow_forward1. Determine the structure of Grignard reagent and carbonyl compound as the starting materials to produce the following alcohols. a) он b) CH2OH c) OH d) Lonarrow_forwardA) nthyl benzene ketone B) ethyl phenyl ketone C) ethane benzene ketone D) ethane phenyl ketone E) phenyl propanonearrow_forward
- Draw the organic products formed when attached allylic alcohol A is treated with following reagent. HCrO4−–Amberlyst A-26 resinarrow_forwardDraw the carbonyl products formed when each alcohol is oxidized with K 2Cr 2O 7.arrow_forwardDraw the products formed when each alcohol is dehydrated with H 2SO 4. Use the Zaitsev rule to predict the major product when a mixture forms.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
