
Concept explainers
Minoxidil is a molecule that causes hair growth in some people. It was originally synthesized as a vasodilator for the treatment of hypertension (high blood pressure). Most of the patients taking the drug for hypertension were seen to grow body hair. Due to other side effects, its oral use was stopped, but it became popular as a topical cream to promote hair growth.
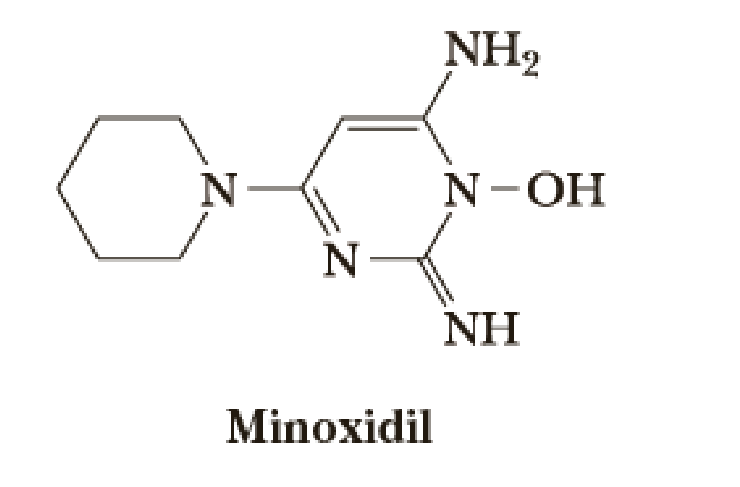
The first key reaction in one synthesis of minoxidil follows. Draw the product of this reaction.
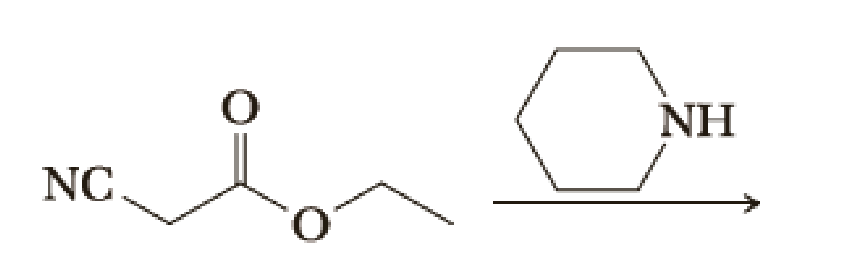
Trending nowThis is a popular solution!

Chapter 18 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry - 4th edition
Living By Chemistry: First Edition Textbook
Inorganic Chemistry
Organic Chemistry - Standalone book
Fundamentals of Heat and Mass Transfer
- PROPOSE A POSSIBLE SYNTHESIS FOR THE GIVEN TRANSFORMATIONarrow_forwardIsoamyl acetate (also known as isopentyl acetate) is an ester that is referred to as “banana flavor” due to its odor that resembles that of banana. It can be synthesized from isopentyl alcohol and acetic acid via nucleophilic acyl substitution mechanism. Propose a mechanism for the synthesis of isoamyl acetate from acetic acid and isoamyl alcohol.arrow_forwardThe anticoagulant Warfarin can be synthesized by a conjugate addition (Michael addition) reaction as shown below: a) Propose a mechanism for the reaction starting from the enolate shown below. You can assume water is present for any proton transfers needed.b) We saw that the keto tautomer is normally preferred over the enol tautomer, however, the enol tautomer is present in the final product (Warfarin) and that is the more stable tautomer in this case. Why do you think the enol tautomer is more stable? Hint: consider the molecule as a whole.arrow_forward
- The following synthesis requires more than one step. Specify the reagents you would use to carry it out.arrow_forward5. Provide the missing reagent indicated by "???": Br ???arrow_forwardDraw a stepwise mechanism for the following reaction, which results in the synthesis of bisphenol F (R = H), an additive used in a variety of packaging materials. Bisphenol F is related to BPA (bisphenol A, R = CH3), a reagent used to harden some plastics, now removed from certain baby products because of its estrogen-like activity that can disrupt endocrine pathways.arrow_forward
- Propose a step by step mechanism for the following reaction.arrow_forwardIn the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or hypnotic. Propose a mechanism for the formation of paraldehyde.arrow_forwardCan you please explain why treatment of 1,4-dihydronaphthalene even a trace of acid results in the formation of 1,2-dihydronaphthalene? I can’t understand it’s mechanism.arrow_forward
- Fluorination of a benzene ring can be accomplished with Selectfluor, a reagent that contains a fluorine bonded to a positively charged nitrogen atom. Fluorination is a useful reaction because several common drugs, such as the cholesterol-lowering drug atorvastatin, contain a fluorine bonded to an aromatic ring. Assuming that fluorination is analogous to other examples of electrophilic aromatic substitution, draw a stepwise mechanism for the following reaction.arrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forwardThe compound eutypine is an antibacterial agent isolated from the fungus Eutypa lata. This fungus results in a disease common to vineyards called eutyposis. Give a sequence of reactions that will take the following reactant and give eutypine when the other reactants used in the sequence are acetylene and acetone.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


