
Concept explainers
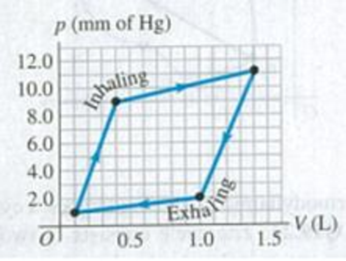
BIO Work Done by the Lungs. The graph in Fig. E19.4 shows a pV-diagram of the air in a human lung when a person is inhaling and then exhaling a deep breath. Such graphs, obtained in clinical practice, are normally somewhat curved, but we have modeled one as a set of straight lines of the same general shape. (Important: The pressure shown is the gauge pressure, not the absolute pressure.) (a) How many joules of net work does this person’s lung do during one complete breath? (b) The process illustrated here is somewhat different from those we have been studying, because the pressure change is due to changes in the amount of gas in the lung, not to temperature changes. (Think of your own breathing. Your lungs do not expand because they’ve gotten hot.) If the temperature of the air in the lung remains a reasonable 20°C, what is the maximum number of moles in this person’s lung during a breath?
Figure E19.4
Learn your wayIncludes step-by-step video

Chapter 19 Solutions
University Physics with Modern Physics (14th Edition)
Additional Science Textbook Solutions
Conceptual Physics (12th Edition)
College Physics (10th Edition)
Essential University Physics (3rd Edition)
Sears And Zemansky's University Physics With Modern Physics
Life in the Universe (4th Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
- You have a particular interest in automobile engines, so you have secured a co-op position at an automobile company while you attend school. Your supervisor is helping you to learn about the operation of an internal combustion engine. She gives you the following assignment, related to a simulation of a new engine she is designing. A gas, beginning at PA = 1.00 atm, VA = 0.500 L, and TA = 27.0C, is compressed from point A on the PV diagram in Figure P19.31 (page 530) to point B. This represents the compression stroke in a fourcycle gasoline engine. At that point, 132 J of energy is delivered to the gas at constant volume, taking the gas to point C. This represents the transformation of potential energy in the gasoline to internal energy when the spark plug fires. Your supervisor tells you that the internal energy of a gas is proportional to temperature (as we shall find in Chapter 20), the internal energy of the gas at point A is 200 J, and she wants to know what the temperature of the gas is at point C. Figure P19.31arrow_forward(a) Given that air is 21% oxygen, find the minimum atmospheric pressure that gives a relatively safe partial pressure of oxygen of 0.16 atm. (b) What is the minimum pressure that gives a partial pressure of oxygen above the quickly fatal level of 0.06 atm? (c) The air pressure at the summit of Mount Everest (8848 m) is 0.334 atm. Why have a few people climbed it without oxygen, while some who have tried, even though they had trained at high elevation, had to tum back?arrow_forwardA pressure versus volume (pv) diagram for a system is shown in the figure. The arrows of the curve indicate the direction of the process, and the points of interest are labeled. The values for the points in the diagram are shown in the table. Volume (m3) Pressure (Pa) v0=27.4 p0=1.00×104 v1=19.3 p1=1.00×104 v2=16.0 p2=4.92×103 v3=13.3 p3=4.92×103 v4=13.3 p4=3.20×103 v5=7.51 p5=1.00×103 Calculate the amount of work done on the system from 0–2 (W02) and then for the entire curve from 0–5 (W05).arrow_forward
- Kristi Yamaguchi is an Olympic gold medalist in figure skating (and Dancing with the Stars champion*). If she has a mass of 42.18 kg and the dimensions of her skate's blade are 4.8 mm by 30.48 cm, what pressure does she exert on the ice? Assume only one skate is down at a time. Provide answer in "atm"arrow_forwardcertain fluid at 12 bar is contained in a cylinder behind a piston, the initial volume being 0.07 m3. Determine the work done by the fluid when it expands reversibly.i. at constant pressure to a final volume of 0.25 m3.ii. According to a law pV3 = constant to a final volume of 0.08 m3.iii. According to a law, p = (A/V2) – (B/V), to a final volume of 0.15 m3and a final pressure of 2 bar, where A and B are constants.arrow_forwardIn a dry-pipe sprinkler valve, the air seat typically has a surface area on which the air pressure acts six times as large as the water seat upon which the water pressure acts. If the water pressure is 105 psi, what air pressure is required to attain a state of equilibrium? answer should be in ( psi )arrow_forward
- Atmospheric pressure atop Mt. Everest is 4 2 3.30x10 N/m . (a) What is the partial pressure of oxygen there if it is 20.9% of the air? (b) What percent oxygen should a mountain climber breathe so that its partial pressure is the same as at sea level, where atmospheric pressure is 1.01 × 10^5 N/m^2 ?arrow_forwardA vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P18.40). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find the height h in Figure P18.40. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder. Figure P18.40arrow_forwardA gas is in a container of volume V0 at pressure P0. It is being pumped out of the container by a piston pump. Each stroke of the piston removes a volume Vs through valve A and then pushes the air out through valve B as shown in Figure P19.74. Derive an expression that relates the pressure Pn of the remaining gas to the number of strokes n that have been applied to the container. FIGURE P19.74arrow_forward
- A vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P16.56). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find die height h in Figure P16.56. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder.arrow_forwardA hand—driven tire pump has a piston with a 2.50cm diameter and a maximum stroke of 30.0 cm. (a) How much work do you do in one stroke if the average gauge pressure is 2.40105N/m2 (about 35 psi)? (b) What average force do you exert on the piston, neglecting friction and gravitational force?arrow_forwardConsider the piston cylinder apparatus shown in Figure P20.81. The bottom of the cylinder contains 2.00 kg of water at just under 100.0c. The cylinder has a radius of r = 7.50 cm. The piston of mass m = 3.00 kg sits on the surface of the water. An electric heater in the cylinder base transfers energy into the water at a rate of 100 W. Assume the cylinder is much taller than shown in the figure, so we dont need to be concerned about the piston reaching the top of the cylinder. (a) Once the water begins boiling, how fast is the piston rising? Model the steam as an ideal gas. (b) After the water has completely turned to steam and the heater continues to transfer energy to the steam at the same rate, how fast is the piston rising?arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





