
Concept explainers
(a)
Interpretation: The number of coordinate bond formed by acetylacetone (acacH)chelating ligand needs to be determined.
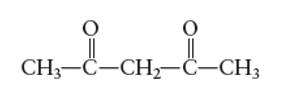
Concept Introduction: A coordination complex is composed of two main parts; metal atom/ ion and ligand. Ligands are the atom/ group of atom/ ion that can donate its extra pair of electrons to the electron deficient metal cation. It leads to the formation of coordinate bond between metal ion and ligand.
(b)
Interpretation: The number of coordinate bond formed by diethylenetriamine chelating ligand needs to be determined.
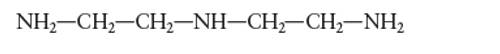
Concept Introduction: A coordination complex is composed of two main parts; metal atom/ ion and ligand. Ligands are the atom/ group of atom/ ion that can donate its extra pair of electrons to the electron deficient metal cation. It leads to the formation of coordinate bond between metal ion and ligand.
(c)
Interpretation: The number of coordinate bond formed by salen chelating ligand needs to be determined.
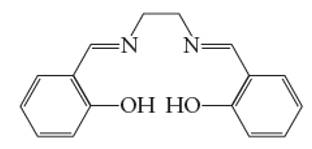
Concept Introduction: A coordination complex is composed of two main parts; metal atom/ ion and ligand. Ligands are the atom/ group of atom/ ion that can donate its extra pair of electrons to the electron deficient metal cation. It leads to the formation of coordinate bond between metal ion and ligand.
(d)
Interpretation: The number of coordinate bond formed by porphine chelating ligand needs to be determined.
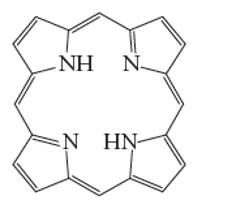
Concept Introduction: A coordination complex is composed of two main parts; metal atom/ ion and ligand. Ligands are the atom/ group of atom/ ion that can donate its extra pair of electrons to the electron deficient metal cation. It leads to the formation of coordinate bond between metal ion and ligand.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Chemical Principles
- Explain how EDTA binds to a heavy metalarrow_forwardWhen dilute HCl is added to zinc pieces taken in a test tube(a) No change takes place(b) the colour of the solution becomes yellow.(c) A pungent smelling gas gets liberated.(d) small bubbles of H2gas appear on the surface of zinc piecesarrow_forwardIs blue color the appearance of resazurin in a reduced state?arrow_forward
- Which of the following statements is true for Tollen's test with acetone? a. Acetone oxidized ammoniacal silver nitrate solution to give a precipitate of silver metal. b. One of the products of the reaction is ammonia. c. The acetone is oxidized to acetic acid. d. Acetone does not react with ammoniacal silver nitrate..arrow_forwardWhich of the following is NOT a dehydrating agent? A) plaster of paris B) phenol C) sodium hypochlorite D) hardening compoundarrow_forward<p>What structural feature do hemoglobin, cytochrome c, and chlorophyll have in common?arrow_forward
- Sodium lauryl sulfate has the same use in embalming fluids as: 1. sodium phosphate 2. citrates 3. sulfonates 4. sodium salt of EDTA 1 & 4 only 3 only 4 only 1,2,3,and 4arrow_forwardPart C (solvents labeled C): How does the number of hydrogen bonding sites within a molecule affect the relative strength of IMF’s?arrow_forwardWhat tests would you use to identify a solution of AG3PO4?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning


