
Concept explainers
(a)
Interpretation: The formula of rutile as TiO2 on the basis of given unit cell needs to be determined.
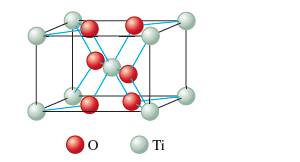
Concept Introduction:
Each solid has a different type of unit cell and crystal lattice that makes it unique from other solids. The atoms are arranged in unit cells.
The unit cells in the solids can arrange in different ways. In a crystal each particle can be represented as spherical atoms which are packed together and bonded to each other equally in all directions. It is called closest packing of crystal that can be in hexagonal cubic packing, close cubic packing, face center cubic packing, etc.
(b)
Interpretation: The oxidation number, for both reactions with identification of oxidizing and reducing agent for each reaction needs to be done.
Concept Introduction:
Each solid has a different type of unit cell and crystal lattice that makes it unique from other solids. The atoms are arranged in unit cells.
The unit cells in the solids can arrange in different ways. In a crystal, each particle can be represented as spherical atoms which are packed together and bonded to each other equally in all directions. It is called closest packing of crystal that can be in hexagonal cubic packing, close cubic packing, face center cubic packing, etc.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Chemical Principles
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





