
CAMPBELL BIOLOGY: CONCEPTS & CONNECTIO
8th Edition
ISBN: 9781323457214
Author: Reece
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 11TYK
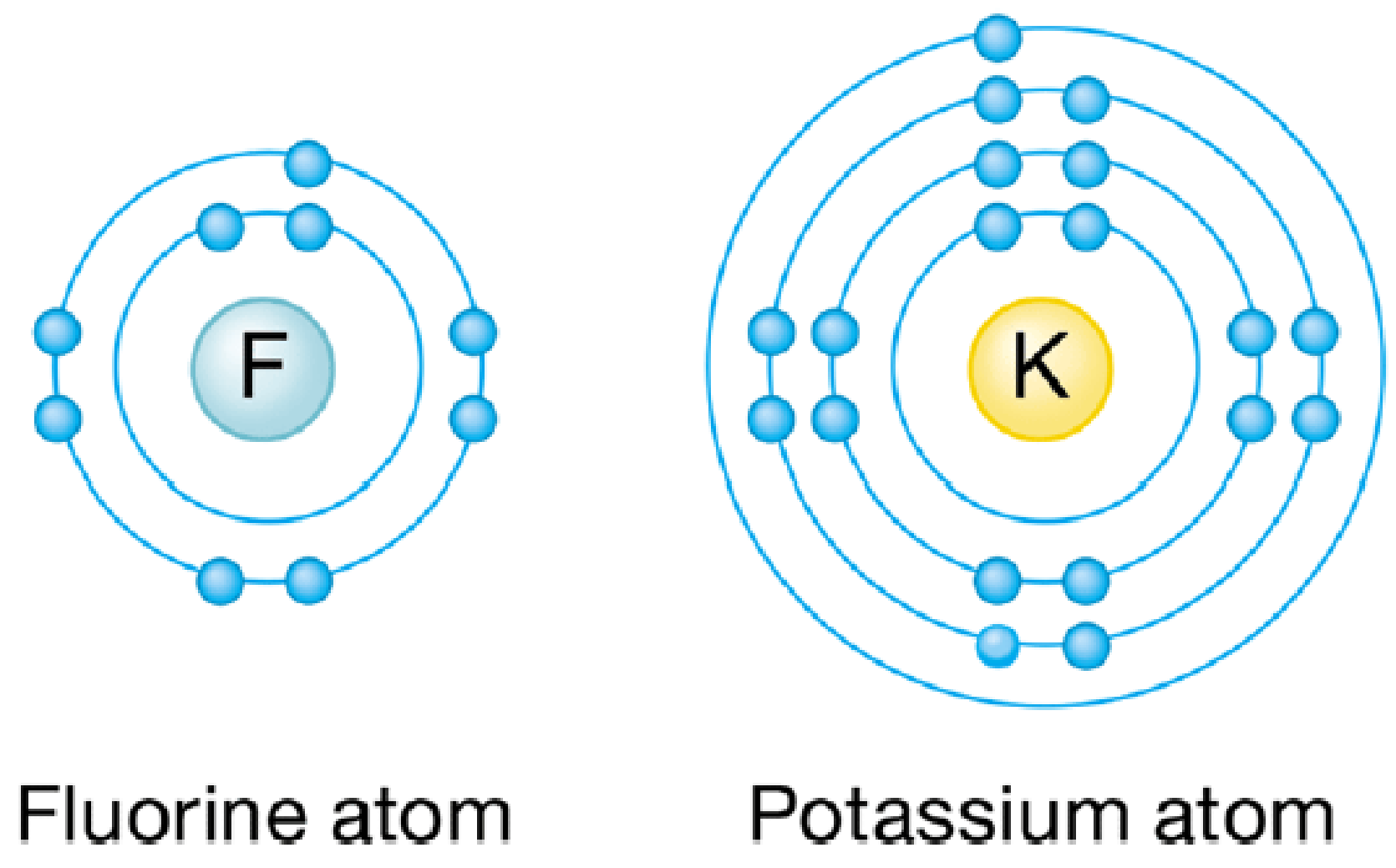
The diagram below shows the arrangement of electrons around the nucleus of a fluorine and a potassium atom. What kind of bond do you think would form between these two atoms?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
CAMPBELL BIOLOGY: CONCEPTS & CONNECTIO
Ch. 2 - Fill in the blanks in this concept map to help you...Ch. 2 - Create a concept map to organize your...Ch. 2 - Changing the _____ would change it into an atom of...Ch. 2 - A solution at pH 6 contains _____H+ than the same...Ch. 2 - Most of the unique properties of water result from...Ch. 2 - A can of cola consists mostly of sugar dissolved...Ch. 2 - Prob. 7TYKCh. 2 - In what way does the need for iodine or iron in...Ch. 2 - Use carbon-12, the most common isotope of carbon,...Ch. 2 - In terms of electron sharing between atoms,...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- What atom has only one proton? a. hydrogen c. a free radical b. an isotope d. a radioisotopearrow_forwardVisit this website (http://openstaxcollege.org/l/ptable) to view the periodic table. In the periodic table of the elements, elements in a single column have the same number of electrons that can participate in a chemical reaction. These electrons are known as valence electrons. For example, the elements in the first column all have a single valence electron—an electron that can be donated in a chemical reaction with another atom. What is the meaning of a mass number shown in parentheses?arrow_forwardWhich type of bond represents a weak chemical bond? a. hydrogen bond b. ionic bond c. covalent bond d. polar covalent bondarrow_forward
- Ionic and covalent bonds join atoms into molecules. What do hydrogen bonds do?arrow_forwardFigure 2.24 Which of the following statements is false? Molecules with the formulas CH3CH2COOH and C3H6O2 could be structural isomers. Molecules must have a double bond to be cis-trans isomers. To be enantiomers, a molecule must have at least three different atoms or groups connected to a central carbon. To be enantiomers, a molecule must have at least four different atoms or groups connected to a central carbon.arrow_forwardThe backbone of organic compounds forms when _________ atoms are covalently bonded.arrow_forward
- Alchemists were the forerunners of modern-day chemists. Many of these medieval scholars and philosophers spent their lives trying to transform lead (atomic number 82) into gold (atomic number 79). Explain why they never succeeded.arrow_forwardFigure 2.12 A pH scale. Here, red dots signify hydrogen ions (H+) and blue dots signify hydroxyl ions (OH). Also shown are the approximate pH values for some common solutions. This pH scale ranges from 0 (most acidic) to 14 (most basic). A change of one unit on the scale corresponds to a tenfold change in the amount of H+ ions. Photos, JupiterImages Corporation. Figure It Out: What is the approximate pH of cola?arrow_forwardAtoms share electrons unequally in an _______ bond. a. ionic c. polar covalent b. hydrogen d. nonpolar covalentarrow_forward
- Magnesium has an atomic number of 12. Which of the following statements is true of a neutral magnesium atom? a. It has 12 protons, 12 electrons, and 12 neutrons. b. It has 12 protons, 12 electrons, and six neutrons. c. It has six protons, six electrons, and no neutrons. d. It has six protons, six electrons, and six neutrons.arrow_forwardOxygen (O) is a(n) _______; the oxygen we breathe (O2) is a(n) _______; and the carbon dioxide we exhale is a(n) _______. a. compound; molecule; element b. atom; compound; element c. element; atom; molecule d. atom; element; molecule e. element; molecule; compoundarrow_forward_______ groups are the acid part of amino acids and fatty acids. a. Hydroxyl (OH) c. Methyl (CH3) b. Carboxyl (COOH) d. Phosphate (PO4)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning


Biology: The Unity and Diversity of Life (MindTap...
Biology
ISBN:9781305073951
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cengage Learning


Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:9781305112100
Author:Cecie Starr, Beverly McMillan
Publisher:Cengage Learning
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license