
Concept explainers
(a)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Base and its conjugate Acid: A Base is a species that can gain a proton. When a base gains a proton
Electron delocalization stabilizes base: If a base has delocalized electrons, then the negative charge that results when the base’s conjugate acid loses a proton will belong to one atom. If a base has delocalized electrons, then the negative charge that results when the base’s conjugate acid loses a proton will be shared by two or more atoms. A base with delocalized electron is more stable than a similar base with localized electrons.
Relative Acid strength: Replacing hydrogen with an electronegative substituent that pulls bonding electrons toward itself increases the strength of the acid.
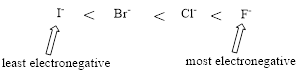
The more electronegative the substituent that replaces hydrogen, the stronger the acid becomes.
Example:
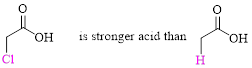
(b)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Base and its conjugate Acid: A Base is a species that can gain a proton. When a base gains a proton
Electron delocalization stabilizes base: If a base has delocalized electrons, then the negative charge that results when the base’s conjugate acid loses a proton will belong to one atom. If a base has delocalized electrons, then the negative charge that results when the base’s conjugate acid loses a proton will be shared by two or more atoms. A base with delocalized electron is more stable than a similar base with localized electrons.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Essential Organic Chemistry, Global Edition
- Which one is more acidic in each pair? And why?arrow_forwardA) for each compound show its conjugate base. lone pairs have been left out. B) rank the conjugate base in the order you would predict, from most to least stable. C) rank the original compounds in order, from strongest to weakest acid.arrow_forwardOChem question regarding acidity Consider ONLY the hydrogen drawn in each compound, list the compounds in order of INCREASING acidity. Also, explain why the most acidic is considered the most acidic and why the least acidic is considered least acidic. Thank you!arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

