
Inorganic Chemistry
5th Edition
ISBN: 9781292134147
Author: Housecroft, Catherine E.
Publisher: Pearson,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 18P
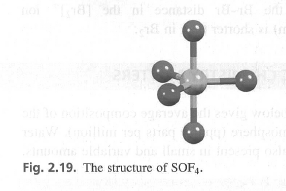
Use the VSEPR model to rationalize the structure of
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Describe the structures of SO2 and SO3 in terms of valence-bond theory.
Write Lewis structures for NF3 and PF5. On the basis of hybrid orbitals, explain the fact that NF3, PF3, and PF5 are stable molecules, but NF5 does not exist.
The molecules SiF4, SF4, and XeF4 have molecular formulas of the type AF4, but the molecules have different molecular geometries. Predict the shape of each molecule, and explain why the shapes differ.
Chapter 2 Solutions
Inorganic Chemistry
Ch. 2 - Draw Lewis structures to describe the bonding in...Ch. 2 - Use the Lewis structure model to deduce the type...Ch. 2 - Draw the resonance structures for the O3 molecule....Ch. 2 - 2.4 Draw Lewis structures for (a) , (b) ,(c) and...Ch. 2 - 2.5 Each of the following is a radical. For which...Ch. 2 - (a) Use VB theory to describe the bonding in the...Ch. 2 - 2.7 Use VB theory and Lewis structure model,...Ch. 2 - 2.8 Does VB theory indicate that the diatomic...Ch. 2 - 2.9 (a) Use MO theory to determine the bond order...Ch. 2 - Prob. 10P
Ch. 2 - Prob. 11PCh. 2 - Draw charge-separated resonance structures to give...Ch. 2 - Prob. 13PCh. 2 - In the following table, match a species in list 1...Ch. 2 - Using the data in table 2.2, determine which of...Ch. 2 - Prob. 16PCh. 2 - 2.17 Use the VSEPR model to predict the structures...Ch. 2 - 2.18 Use the VSEPR model to rationalize the...Ch. 2 - Determine the shapes of each of the following...Ch. 2 - 2.20 State whether you expect the following...Ch. 2 - 2.21 (a) Draw resonance structure for the CO,...Ch. 2 - Prob. 22PCh. 2 - Prob. 23PCh. 2 - Suggest reasons for the following observations....Ch. 2 - Prob. 25PCh. 2 - Prob. 26PCh. 2 - 2.27 (a) Write down the ions that are present in...Ch. 2 - 2.28 Assuming that VSEPR model can be applied...Ch. 2 - Critically compare the VB and MO treatments of the...Ch. 2 - The table below gives the average composition of...Ch. 2 - Carbon monoxide is a toxic pollutant which arises...Ch. 2 - 2.32 Volcanoes and deep sea hydrothermal vents are...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Could the anion Li2 exist? What is the ions bond order?arrow_forwardIn each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardExplain in terms of bonding theory why all four hydrogen atoms of allene, H2CCCH2, cannot lie in the same plane.arrow_forward
- It is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forwardA student claims that if element D is in group 18, then the molecular geometry of DF4(g) is square planar. Do you agree or disagree with the student? Justify your answer in terms of the VSEPR theory.arrow_forwardPredict the ordering, from shortest to longest, of the bondlengths in CO, CO2, and CO32 - .arrow_forward
- Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicated atoms.arrow_forwardIs the electron density consistentwith equal contributions from the tworesonance structures for O3? Explain.arrow_forwardThe main component of air is N2. From the molecular orbital diagram of N2, predict its bond order and whether it is diamagnetic or paramagnetic.arrow_forward
- The bond order between C and O in CO+is 3.5 but bond order between N and O in NO+is three only. Explain why?arrow_forwardDraw the Lewis structure of SOF₄ (with minimized formal charges) and then determine the hybridization of the central atom.arrow_forwardWrite the ground-state electron configuration of O2 and calculate the bond order.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY