
Interpretation:
The group should be identified when the valance shell configuration is
Concept introduction:
The periodic table is given below,
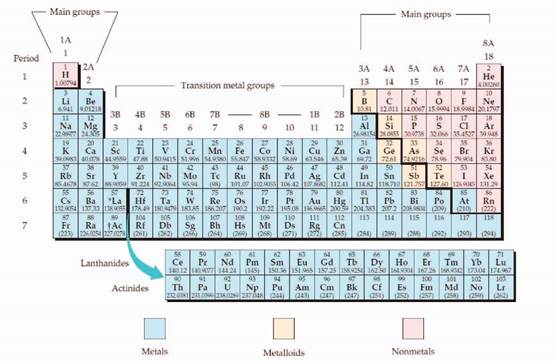
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. Atom consists of s, p, d, and f orbitals.
The Aufbau principle:
The Aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Modified Mastering Chemistry With Pearson Etext -- Standalone Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry (8th Edition)
- Arrange in order of increasing nonmetallic character (a) the period 3 elements Na, Cl, & Mg (b) the Group 7A elements At, F, & Iarrow_forwardA principal shell with a value of n=3 would contain s,p,d, and f orbitals true or falsearrow_forwardWhat is the empirical formula of a compound that contains 72.0% carbon, 12.0% hydrogen and 16.0% oxygen by mass?arrow_forward
- What three things did the bohr model add to our understanding of atomic structure?arrow_forwardHow many oxygen atoms are in this molecular formula? 3H2 Oarrow_forwardA compound with empirical formula C2H5O was found in a separate experiment to have a molar mass of approximately 90 g. What is the molecular formula of the compound?arrow_forward
- How many electrons are in the outer shell of each of the following atoms?arrow_forwardIdentify the names of the following structure. (with alpha/beta and L-D designation)arrow_forwardIf one compound has the formula C5H10 and another has the formula C4H10, are the two compounds isomers? Explain.arrow_forward

 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

