
Interpretation:
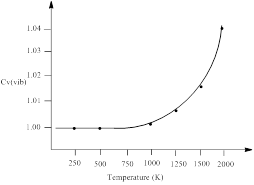
H2 gas has a single vibrational frequency of 1.295 x 1014 s-1. The vibrational contribution to the heat capacity, Cv (vib) versus temperature from 0 to 2000 K is to be plotted.
Concept introduction:
Generally, the vibrational partition function of a molecule is derived by incorporating the calculated vibrational energy values into the exponentials and found in notation of Cv (vib) and adding everything. heat capacity (thermal capacity) is the quantity of heat required to raise the temperature of the system from the lower limit to higher divided by the temperature difference of the system. When the mass of the system is taken as 1gram, the heat capacity is denoted as specific heat capacity. Similarly, when the mass of the system taken as 1 mole, the heat capacity is referred as molar heat capacity. Heat capacity is generally described as the symbol C. Mathematically, the heat capacity of the system between two temperature T1 and T2 can be expressed as
Answer to Problem 2.92E
The vibrational partition function is given by Cv >(vib) =
| Temperature (K) | Cv (vib) |
| 100 | ≈1 |
| 500 | ≈1 |
| 1000 | 1.00202 |
| 1500 | 1.016 |
| 2000 | 1.047 |
Explanation of Solution
Given,
The vibrational partition function is given by Cv (vib) =
Single vibrational frequency of hydrogen = 1.295 x 1014 s-1
We know that,
• Cv (vib) at 100 K:
Cv (vib) ≈1
• Cv (vib) at 500 K:
Cv (vib) ≈1
• Cv (vib) at 1000 K:
Cv (vib) = 1.002
• Cv (vib) at 1500 K:
Cv (vib) = 1.016
• Cv (vib) at 2000 K:
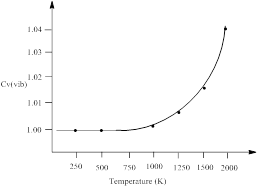
| Temperature (K) | Cv (vib) |
| 100 | ≈1 |
| 500 | ≈1 |
| 1000 | 1.00202 |
| 1500 | 1.016 |
| 2000 | 1.047 |
Thus, the vibrational contribution to the heat capacity, Cv (vib) versus temperature from 0 to 2000 K is plotted.
Want to see more full solutions like this?
Chapter 2 Solutions
Physical Chemistry
- What are the numerical values of the heat capacities c-v and c-p of a monatomic ideal gas,in units of cal/mol.K and L.atm/mol.K?arrow_forwardWhat is the finaltemperature of0.122 mole ofmonatomic ideal gas that performs 75J of work adiabatically if the initial temperature is 235C?arrow_forwardCalculate the work for the isothermal, reversible compressionof 0.245 moleof an idealgas going from 1.000L to 1.00 mL if the temperature were 95.0C.arrow_forward
- Determine an expression for V/T p, n in terms of and . Does the sign on the expression make sense in terms of what you know happens to volume as temperature changes?arrow_forwardA 1.00 mol sample of H2 is carefully warmed from 22 K to 40 K at constant volume. a What is the expected heat capacity of the hydrogen? b What is q for the process?arrow_forwardA sample of an ideal diatomic gas is compressed adiabatically and reversibly to double its initial pressure. By what percentage does its absolute temperature change in a the low-temperature limit and b the high-temperature limit?arrow_forward
- The Dieterici equation of state for one mole of gas is p=RTe-aVRTV-b Where a and b are constants determined experimentally. For NH3g, a = 10.91 atm. L2 and b = 0.0401 L. Plot the pressure of the gas as the volume of 1.00 mol of NH3g expands from 22.4 L to 50.0 L at 273 K, and numerically determine the work done by the gas by measuring the area under the curve.arrow_forwardIf the heat capacity varies withtemperature, abetter form ofequation 2.9 isto solve q=TiTfnCT-t A 50.0-g sample of white phosphorus is heated from 298 K to 350K. If its molar heat capacity is CT- = 56.990.1202T J/mol.K, how much heat is needed?arrow_forwardA 220-ft3 sample of gas at standard temperature and pressure is compressed into a cylinder, where it exerts pressure of 2000 psi. Calculate the work (in J) performed when this gas expands isothermally against an opposing pressure of 1.0 atm. (The amount of work that can be done is equivalent to the destructive force of about 1/4 lb of dynamite, giving you an idea of how potentially destructive compressed gas cylinders can be if improperly handled!)arrow_forward
- For CO_2 how would the theoretical ratio of C_p (heat capacity at constant pressure)/ C_v (heat capacity at constant volume) be affected if the molecule were nonlinear, such as SO_2, instead of linear? Could you decide between these two structures from the C_p/C_v ratio alone?arrow_forwardWhat is the reversible work, in joules, for the expansion of 2.50 mol of an ideal gas from 4.7 bar to 1.65 bar at a constant temperature of 298 K.arrow_forwardCalculate the theoretical value of C_p (constant pressure heat capacity)/ C_v (constant volume heat capacity) for the following gases: Ar, CO_2, and N_2arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



