
BIOLOGY
12th Edition
ISBN: 9781260169614
Author: Raven
Publisher: RENT MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 2A
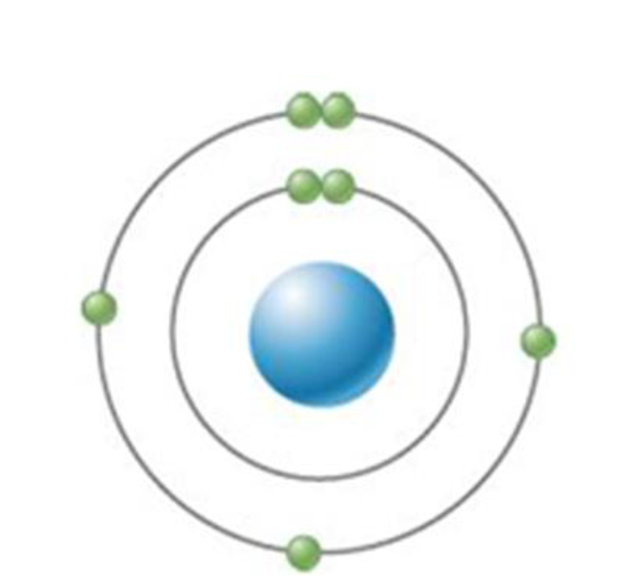
Refer to the element pictured. How many covalent bonds could this atom form?
a. Two
b. Three
c. Four
d. None
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is an alloy?
A. A compound of two metals
B. An element of two metals
C. A mixture of two metals
D. A molecule of two metals
Which element is a metal?
A. C
B. Rn
C. CI
D. Cr
Which statement is true of all atoms that are anions?(A) The atom has more electrons than protons.(B) The atom has more protons than electrons.(C) The atom has fewer protons than does a neutral atomof the same element.(D) The atom has more neutrons than protons.
Chapter 2 Solutions
BIOLOGY
Ch. 2.1 - Define an element based on its composition.Ch. 2.1 - Describe the relationship between atomic structure...Ch. 2.1 - Explain where electrons are found in an atom.Ch. 2.2 - Relate atomic structure to the periodic table of...Ch. 2.2 - Prob. 2LOCh. 2.3 - Prob. 1LOCh. 2.3 - Prob. 2LOCh. 2.3 - Prob. 3LOCh. 2.4 - Relate how the structure of water leads to...Ch. 2.4 - Prob. 2LO
Ch. 2.5 - Prob. 1LOCh. 2.5 - Prob. 2LOCh. 2.5 - Prob. 3LOCh. 2.6 - Prob. 1LOCh. 2.6 - Prob. 2LOCh. 2 - Data analysis If we call each step on the x-axis...Ch. 2 - The property that distinguishes an atom of one...Ch. 2 - If an atom has one valence electronthat is. a...Ch. 2 - An atom with a net positive charge must have more...Ch. 2 - The isotopes carbon-12 and carbon-14 differ in a....Ch. 2 - Which of the following is NOT a property of the...Ch. 2 - Ionic bonds arise from a. shared valence...Ch. 2 - A solution with a high concentration of hydrogen...Ch. 2 - Using the periodic table on page 22, which of the...Ch. 2 - Refer to the element pictured. How many covalent...Ch. 2 - Prob. 3ACh. 2 - Prob. 4ACh. 2 - If you shake a bottle of oil and vinegar then let...Ch. 2 - The decay of radioactive isotopes involves changes...Ch. 2 - Prob. 1SCh. 2 - A popular theme in science fiction literature has...Ch. 2 - Prob. 3S
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- What atom has only one proton? a. hydrogen c. a free radical b. an isotope d. a radioisotopearrow_forwardWhich type of bond represents a weak chemical bond? a. hydrogen bond b. ionic bond c. covalent bond d. polar covalent bondarrow_forwardSelect the sentence that correctly describes the relationship between this pair of molecules. a They are both enantiomers and isomers. b They are enantiomers but not isomers. c They are isomers but not enantiomers. d They are neither enantiomers nor isomers.arrow_forward
- Draw and label a simplified model of an atom. Explain how this model misrepresents our understanding of atomic structurearrow_forwardWhich bond in a polypeptide backbone does not freely rotate? a. The bond between CO and NH b. The bond between C-alpha and NH c. The bond between C-alpha and CO d. The bond between C-alpha and C-beta c. d.arrow_forwardWhich is true about carbon atoms? a. Each of the orbitals in the second shell has one electron in it. b. Only the 2p orbitals have unpaired electrons c. Carbon has high electronegativity d. Carbon binds hydrogen with polar covalent bondsarrow_forward
- Refer to the following figure (first three rows of the periodic table) to answer the questions below. What element does not react with other elements? Group of answer choices A. hydrogen B. helium C. oxygen D. siliconarrow_forwardThe atomic number gives thea. number of neutrons in the nucleus.b. number of protons in the nucleus.c. weight of the atom.d. number of protons in the valence shell.arrow_forwardhow many hydrogen can be formed from the question abovearrow_forward
- Which of the following items are pure elements—that is, made up of only one type of atom? Check all that apply. A. Iron B. Cake C. Carbonarrow_forwardThe atomic mass of an element can be used to determine A) the chemical properties of the element B) the number of protons in the element C) the number of neutrons in the element D) the number of protons plus neutrons in the element E) both the number of protons and the chemical properties of the elementarrow_forwardCompare an element and a compound.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College




Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license