
Concept explainers
What reagent is needed to convert
a. 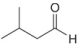 b.
b. 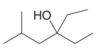 c.
c. 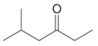 d.
d. 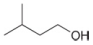
(a)
Interpretation: The reagent needed to convert
Concept introduction: Acid chlorides, which contains good leaving group
Answer to Problem 20.30P
The reagent needed to convert
Explanation of Solution
The given compound is aldehyde. Acid chlorides on reaction with mild reducing agent like lithium tri-tert-butoxyaluminium hydride
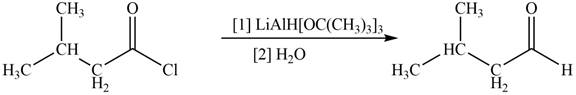
Figure 1
Therefore,
The reagent needed to convert
(b)
Interpretation: The reagent needed to convert
Concept introduction: Acid chlorides, which contains good leaving group
Answer to Problem 20.30P
The reagent needed to convert
Explanation of Solution
The structure of given ketone compound shows that the hydroxyl group is attached to tertiary carbon atom. Acid chlorides on reaction with two equivalents of Grignard reagent convert into tertiary alcohol. The first equivalent of Grignard reagent remove good leaving
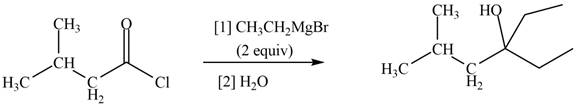
Figure 2
Therefore,
The reagent needed to convert
(c)
Interpretation: The reagent needed to convert
Concept introduction: Acid chlorides, which contains good leaving group
Answer to Problem 20.30P
The reagent needed to convert
Explanation of Solution
The structure of given compound shows that the carbonyl carbon is attached to ethyl group. Ethyl group can occupy the position of leaving group
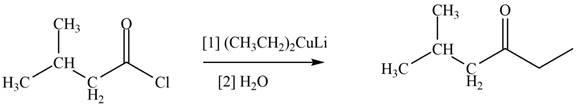
Figure 3
Therefore,
The reagent needed to convert
(d)
Interpretation: The reagent needed to convert
Concept introduction: Acid chlorides, which contains good leaving group
Answer to Problem 20.30P
The reagent needed to convert
Explanation of Solution
Acid chlorides on reaction with strong reducing agent like lithium aluminium hydride
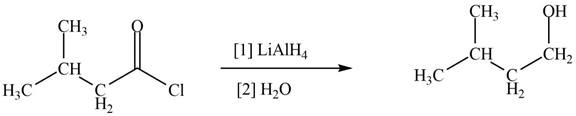
Figure 4
Therefore,
The reagent needed to convert
Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY
- Give the IUPAC name for each compound. CH3 a. CH,CHCH-C CHO b. CH3 H CH2CH3arrow_forward3. What product is formed when HO(CH₂) CHO is treated with each reagent? c. Ag₂O, NH OH d. Na₂Cr₂O₂, H₂SO4, H₂O a. NaBH, CH₂OH b. PCC 400arrow_forwardb. a. C. HO Draw the products of each reaction. COOH COOH + NaOCH3 OH OH H₂SO4 H₂SO4arrow_forward
- 1. Draw the products in each reaction. a. H b. CH3-C-CH₂CH3 CI -OH K+ -OC(CH3)3 CH₂CH3 a Xa C. d. CH₂CH₂OH CH3CH₂O™arrow_forwardIndicate the product obtained by reacting A and B. A || H3C-S-CH₂ Na* || CH 3 B CH2. C. CH3 H3C CH3arrow_forward2. What is the product(s) of the following reaction? A HN B. N A NH2 [H+] -H₂O C. D. HN 3. What is/are the product(s) of the following reaction? A. B. D [H+] -H₂O C.arrow_forward
- 7. Draw the products formed when each compound is trea ted with HNO3 and H2SO4. OCH, OCH, CI CH3 NO2 Br a. b. с. d. Br ČOOCH3arrow_forwardSynthesize each compound from acetylene. You may use any other organic or inorganic reagents. C. CH,CH,CH,CH, CHO d. CH3CH, CHỖ CHarrow_forwardCompound that is most easily hydrolyzed by acid in water `NH A. В. F C. D.arrow_forward
- Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridinearrow_forward• Whał arc the IUPAC namar of the ff. Carboxylic acias? a. COOH COOH CH3 b. I f. .COOH CHJCH2 ÇHCHCOOH CH3 NO2 tON C. COOH ноос d. COOHarrow_forwardWhat alkenes are formed when each alcohol is dehydrated with TSOH? Label the major product when a mixture results. OH Xom OH a. OH b. CH₂CH3 OH C. d. CH₂CH₂CH₂CH₂OH e.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





