
Concept explainers
Interpretation:
The structural formula of the product formed in the given Diels-Alder reaction, has to be drawn with stereochemistry.
Concept Introduction:
Diels-Alder reaction:
It is the reaction of conjugated dienes with double or triple bonded compounds which are known as “dienophiles”. The reaction is a
Example:
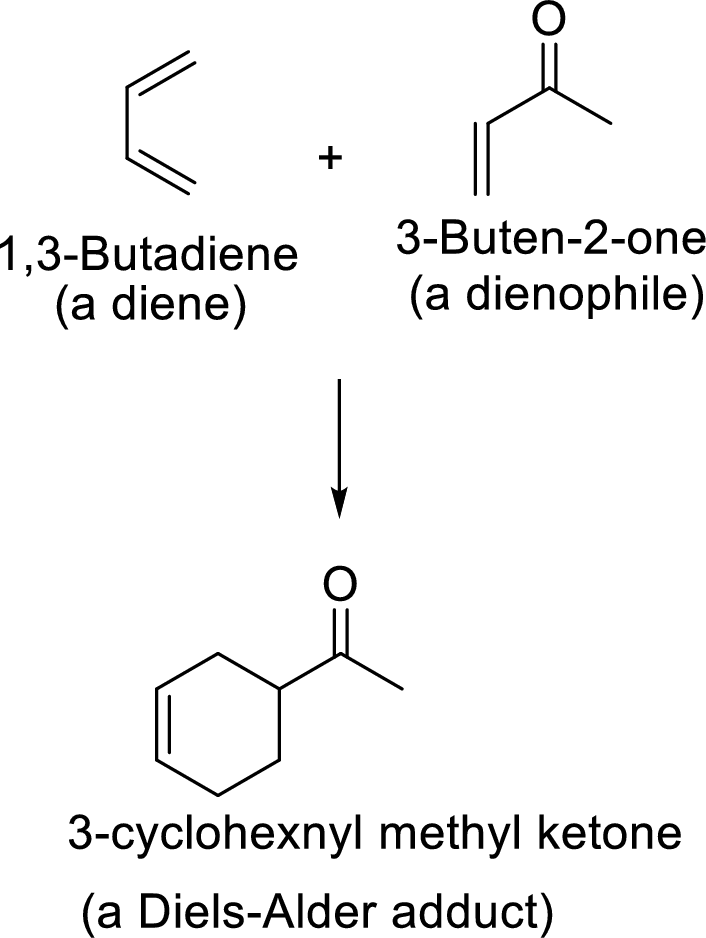
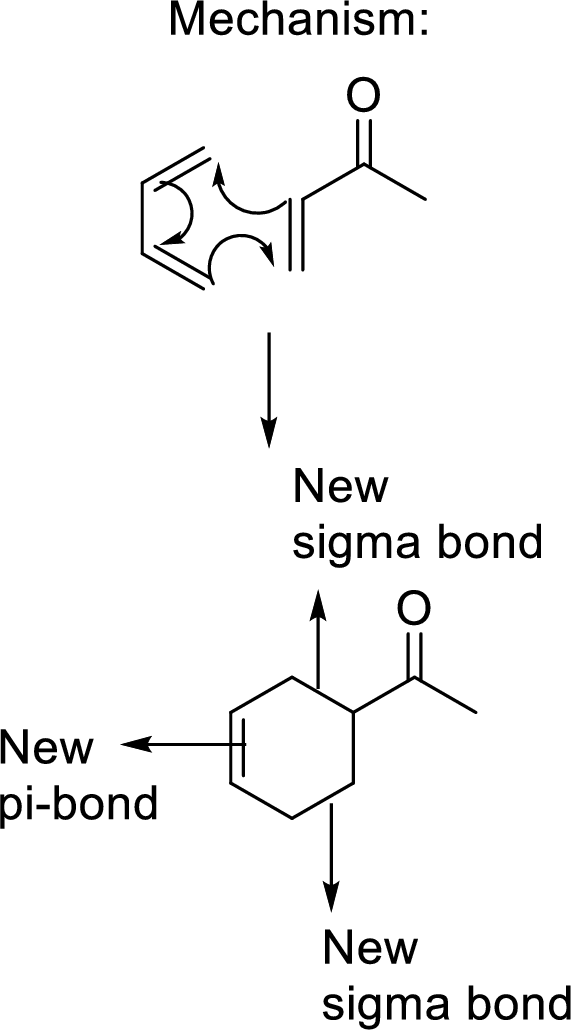
This mechanism shown that three
Stereochemistry in a product formed: In the product of a
Racemic mixture in Diels-alder reaction:
In the Diels-alder reaction, the formation of two new sigma bonds results in the formation of two new chiral centres that are enantiomers to each other. So, the Diels-alder adduct is the mixture of two enantiomers and therefore it is being a racemic Diels-alder adduct.
Trending nowThis is a popular solution!

Chapter 20 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- A chemist is attempting to synthesize a complex natural product with a highly strained cyclohexene ring system. Which type of reactants would be most suitable for achieving this goal, and why? Provide a detailed explanation of the choice of reactants and the expected outcome in terms of the Diels-Alder reaction.arrow_forwardDraw a structural formula for the product of this Diels-Alder reaction, including all stereoisomers of the productarrow_forwardPredict the products of the following Diels-Alder reaction.arrow_forward
- Explain why the Diels Alder reaction would have synthetic significance in the production of biologically active compounds.arrow_forwardPredict the product(s) of the following Diels-Alder reaction. Include stereochemistry if appropriate.arrow_forwardPlease show all arrow pushingj mechaaisais. Thank you!arrow_forward
- Predict the major product for the Diels-Alder reaction.arrow_forwardFollowing is an example of a type of reaction known as a Diels-Alder reaction 1,3-Pentadiene Ethylene 3-Methylcyclohexene (a racemic mixture) The Diels-Alder reaction between a diene and an alkene is quite remarkable in that it is one of the few ways that chemists have to form two new carbon-carbon bonds in a single reaction. Given what you know about the relative strengths of carbon-carbon sigma and pi bonds, would you predict the Diels-Alder reaction to be exothermic or endothermic? Explain your reasoning.arrow_forwardStep 6: Now that you have determined the substrates and mechanism of a Diels-Alder reaction, you will learn how to recognize when you should use the Diels-Alder reaction. In a synthesis reaction, if you are given a cyclohexene product with no other obvious functional group transformations and an electron-withdrawing group two carbons away from the alkene, it is likely made via the Diels-Alder reaction. Deduce the structures of the starting materials to form the Diels-Alder adduct shown. ..... CN CN Diene + Dienophilearrow_forward
- Under certain conditions, 1,3-butadiene can function as both a diene and a dienophile. Draw a structural formula for the Diels-Alder adduct formed by reaction of 1,3-butadiene with itself.arrow_forwardThe endo rule in the Diels-Alder reaction predicts the formation of a bicyclic product. However, in many cases the endo compound is not the thermodynamic product. Briefly elaborate why this is the case.arrow_forwardWrite a general rule that can be used to predict the major product of a Diels–Alder reaction between an alkene with an electron-withdrawing substituent and a diene with a substituent that can donate electrons by resonance depending on the location of the substituent on the diene.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

