
Draw the products formed when pentanal
a.
b.
c.
d.
e.
f.
g.
h.
i.
j.
k.
l. The [product in (a), then TBDMS-Cl, imidazole.
(a)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: The substitution reaction involves the replacement of one functional group by other functional group. In nucleophilic substitution an electron rich species attack the species that is deficient in electrons. The electrophile and the leaving group together form a substrate. The nucleophile attacks over the substrate and there occurs the removal of leaving group from the substrate.
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
![]()
Figure 1
Explanation of Solution
The reaction that shows the formation of product when pentanal reacts with
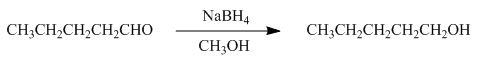
Figure 2
In the given reaction, sodium borohydride is used as a reducing agent. Sodium borohydride is used to for the reduction of carbonyl compounds to alcohols.
The product formed by the reaction of pentanal with
(b)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: The substitution reaction involves the replacement of one functional group by other functional group. In nucleophilic substitution an electron rich species attack the species that is deficient in electrons. The electrophile and the leaving group together form a substrate. The nucleophile attacks over the substrate and there occurs the removal of leaving group from the substrate.
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
![]()
Figure 1
Explanation of Solution
The reaction that shows the formation of product when pentanal reacts with
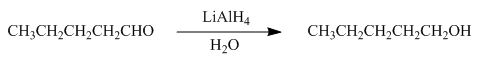
Figure 3
In the given reaction,
The product formed by the reaction of pentanal with
(c)
Interpretation: The product formed by the reaction of pentanal with the
Concept introduction: Addition of hydrogen takes place across a double bond of alkenes to form alkanes. This process is known as hydrogenation. It takes place in the presence of catalysts such as nickel, platinum or palladium.
Answer to Problem 20.38P
The product formed by the reaction of pentanal with the
![]()
Figure 1
Explanation of Solution
The reaction that shows the formation of product when
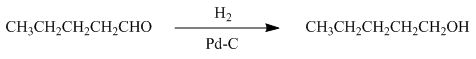
Figure 4
In the given reaction, hydrogenation of alkene takes place in the presence of palladium.
The product formed by the reaction of pentanal with the
(d)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction:
Answer to Problem 20.38P
No product is formed by the reaction of pentanal with
Explanation of Solution
The product formed by the reaction of pentanal with
![]()
Figure 5
No product is formed by the reaction of pentanal with
(e)
Interpretation: The product formed by the reaction of pentanal with the
Concept introduction: Organometallic reagents like
Answer to Problem 20.38P
The product formed by the reaction of pentanal with the
![]()
Figure 6
Explanation of Solution
The product formed by the reaction of pentanal with the
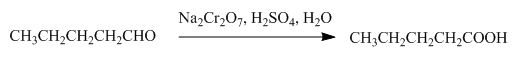
Figure 7
The product formed by the reaction of pentanal with the
(f)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction:
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
![]()
Figure 6
Explanation of Solution
The product formed by the reaction of pentanal with
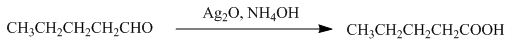
Figure 8
The product formed by the reaction of pentanal with
(g)
Interpretation: The product formed by the reaction of pentanal with the
Concept introduction: Grignard reagent is prepared by the reaction of alkyl or aryl bromide with magnesium metal in the presence of ether. The reaction of Grignard reagent with an aldehyde/ketone followed by hydrolysis yields an alcohol.
Answer to Problem 20.38P
The product formed by the reaction of pentanal with the
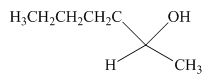
Figure 9
Explanation of Solution
The reaction that shows the formation of product when
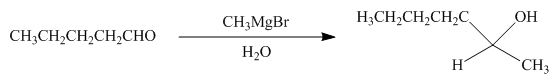
Figure 10
In the given reaction, Grignard reagent is used. This reagent attacks on the carbonyl compound to form alkoxides. In the next step, protonation with water takes place to give the final product.
The product formed by the reaction of pentanal with the
(h)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: Organometallic reagents like
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
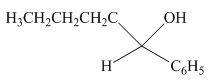
Figure 11
Explanation of Solution
The reaction that shows the formation of product when
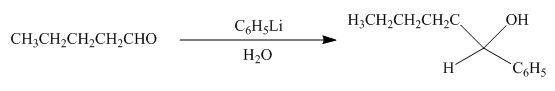
Figure 12
The product formed by the reaction of pentanal with
(i)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: Organometallic reagents like
Answer to Problem 20.38P
No product formed by the reaction of pentanal with
Explanation of Solution
The product formed by the reaction of pentanal with
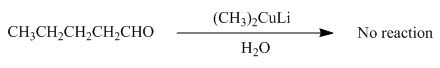
Figure 13
Aldehydes do not react with organo copper reagent because these reagents are less reactive and cannot attack to the electrophilic centre of the aldehyde group.
(j)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: Organometallic reagents like
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
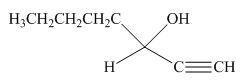
Figure 14
Explanation of Solution
The product formed by the reaction of pentanal with
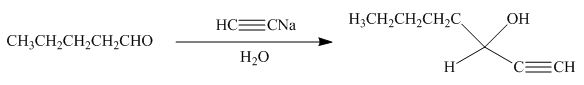
Figure 15
(k)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: Organometallic reagents like
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
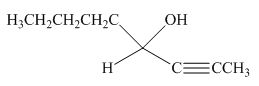
Figure 16
Explanation of Solution
The reaction that shows the formation of product when
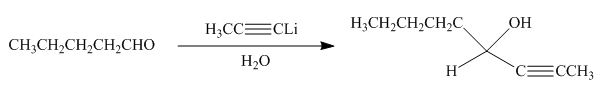
Figure 17
(l)
Interpretation: The product formed by the reaction of pentanal with
Concept introduction: Organometallic reagents like
Answer to Problem 20.38P
The product formed by the reaction of pentanal with
Figure 18
Explanation of Solution
The product formed by the reaction of pentanal with
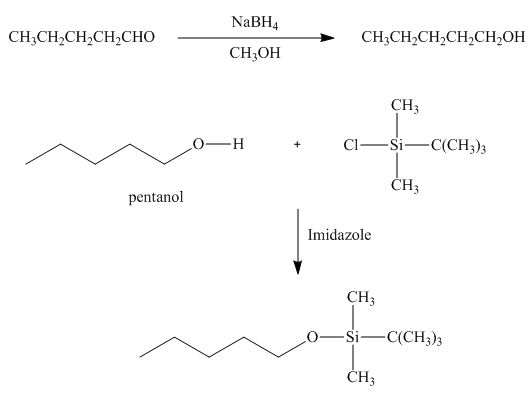
Figure 19
In the given reaction, sodium borohydride is used as a reducing agent. Sodium borohydride is used to for the reduction of carbonyl compounds to alcohols.
The product formed by the reaction of pentanal with the
Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY (LOOSELEAF) >CUSTOM<
- Draw the products formed when each alcohol is dehydrated with H 2SO 4. Use the Zaitsev rule to predict the major product when a mixture forms.arrow_forwardwhich organic compound dissolves in water? 1. 2-pentanol 2. 1-hexanol 3. diethylether 4. 3-methyl-3-pentanol which organic compound dissolves in 10% NaOH? 1. 2-pentanol 2. 1-hexanol 3. diethylether 4. 3-methyl-3-pentanolarrow_forwardHow to draw 2 propanolarrow_forward
- Draw the product formed when the alcohol cyclobutanol is dehydrated with H2SO4.arrow_forwardGive a systematic (IUPAC) name for each diol. ) HO¬(CH2)8¬OHarrow_forward1. Which alcohol has a higher boiling point?a. (i) 2-methylpropan-2-ol or (ii) butan-2-olb. (i) hexan-1-ol or (ii) 3,3-dimethylbutan-1-olarrow_forward
- Molecule Type Boiling point (°C) CH3CH2CH3 Alkane -42 CH3CHO Aldehyde +21 CH3CH2OH Alcohol +78 i. Why is the boiling point of the aldehyde greater than that of the alkane?ii. Why is the boiling point of alcohol the highest?iii. Explain why the solubility of aldehydes and alcohols falls as the molecules get bigger.arrow_forwardShow how each alcohol or diol can be prepared from an alkene. (a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol (d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol (g) 1,2-Hexanediolarrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forward
- Give a systematic (IUPAC) name for each diol.(a) CH3CH(OH)(CH2)4CH(OH)C(CH3)3 (b) HO¬(CH2)8¬OHarrow_forwardShow how each alcohol or diol can be prepared from an alkene.(a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol(d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol(g) 1,2-Hexanedioarrow_forwardWhat kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholsarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


