
Concept explainers
Treatment of isobutene
that reacts with
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry
- Ketones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm-1 and M+=74 in the mass spectrum?arrow_forwardAddition of m-xylene to the strongly acidic solvent HF/SbF5 at 45C gives a new species, which shows 1H-NMR resonances at 2.88 (3H), 3.00 (3H), 4.67 (2H), 7.93 (1H), 7.83 (1H), and 8.68 (1H). Assign a structure to the species giving this spectrum.arrow_forward3-Chlorocyclopropene, on treatment with AgBF4, gives a precipitate of AgCl and a stable solution of a product that shows a single 1H NMR absorption at 11.04 δ. What is a likely structure for the products, and what is its relation to HĂ¼ckel’s rule?arrow_forward
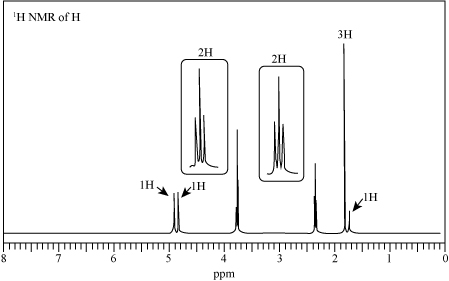
- Nitriles, R–=C≡N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propanenitrile, CH3CH2C≡N, if it has IR absorptions from 2500–3100 cm-1 and at 1710 cm-1, and has M+=74?arrow_forwardReaction of butanenitrile (CH3CH2CH2CN) with methylmagnesium bromide (CH3MgBr), followed by treatment with aqueous acid, forms compound G. G has a molecular ion in its mass spectrum at m/z = 86 and a base peak at m/z = 43. G exhibits a strong absorption in its IR spectrum at 1721 cm−1 and has the 1H NMR spectrum given below. What is the structure of G? We will learn about the details of this reaction in Chapter 22.arrow_forwardReaction of butanenitrile (CH3CH2CH2CN) with methylmagnesium bromide (CH3MgBr), followed by treatment with aqueous acid, forms compound G. G has a molecular ion in its mass spectrum at m/z = 86 and a base peak at m/z = 43. G exhibits a strong absorption in its IR spectrum at 1721 cm−1 and has the 1H NMR spectrum given below. What is the structure of G?arrow_forward
- Ethers are not easily differentiated by their infrared spectra, but they tend to form predictable fragments in the mass spectrum. The following compounds give similar butdistinctive mass spectra.O Obutyl propyl ether butyl isopropyl etherBoth compounds give prominent peaks at m>z 116, 73, 57, and 43. But one compoundgives a distinctive strong peak at 87, and the other compound gives a strong peak at 101.Determine which compound gives the peak at 87 and which one gives the peak at 101.Propose fragmentations to account for the ions at m>z 116, 101, 87, and 73.arrow_forwardCyclohexene has the formula C6H1o and the structure shown in Figure 4-4. When cyclohexene is treated with acid and water it forms Compound A with the formula C6H12O. When Compound A is treated with an oxidizing agent, it forms Compound B with the formula C6H100. IR spectra for Compounds A and B are shown in Figure 4-3. The best structure for A is The best for B isarrow_forwardTreatment of 2-methylpropanenitrile [(CH3)2CHCN] with CH3CH2CH2MgBr, followed by aqueous acid, affords compound V, which has molecular formula C7H14O. V has a strong absorption in its IR spectrum at 1713 cm−1, and gives the following 1H NMR data: 0.91 (triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and 2.60 (septet, 1 H) ppm. What is the structure of V? We will learn about this reaction in Chapter 20.arrow_forward
- N-propylbenzene, C6H5CH2CH3, contains C (sp3) -H and C (sp2) -H bonds. Its IR spectrum shows strong or medium absorptions at 3085, 3064, 3028, 2960, 2931 and 2873 cm ^ -1, as well as bands below 1600cm -1. Which statement is wrong? A.) Stretching of the C (sp3) -H bonds results in absorptions at lower wave numbers than the stretching of the C (sp2) -H bonds. B.) The absorptions at 2960, 2931 and 2873 cm ^ -1 are assigned to stretching of the C (sp3) -H bonds. C.) The absorptions at 3085, 3064 and 3028 cm ^ -1they are assigned to stretching of the C (sp2) -H bonds. D.) Each absorption can be assigned to the stretch mode of a particular bond in the propylbenzene molecule.arrow_forwardThe mass spectrum of 1-ethyl-1-methylcyclohexane shows many fragments, with two in very large abundance. Kne appears af m/z=111 and the other appears at m/z=97. Identify the structure of each of these fragments.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

