
Organic Chemistry
11th Edition
ISBN: 9781118147399
Author: Solomons, T. W.
Publisher: John Wiley & Sons Inc
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 2Q
Which would yield propylamine?
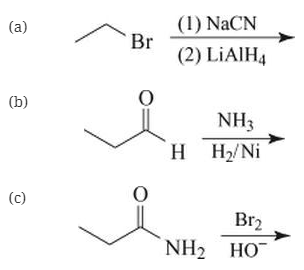
(d) Two of the above
(e) All of the above
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please answer all. Supply the missing detail for the following reactions. Please explain
Draw the structures of the following compounds. (a) isobutyl bromide
Which of the following would deprotonate the thiol in the reaction below? Please look at pic to help answer question
(A) NaH
(B) NaOH
(C) KOH
(D) All the above
(E) None of the Above
Chapter 20 Solutions
Organic Chemistry
Ch. 20 - Prob. 1PPCh. 20 - Prob. 2PPCh. 20 - Prob. 3PPCh. 20 - PRACTICE PROBLEM 20.5 Outline a preparation of...Ch. 20 - Prob. 5PPCh. 20 - Prob. 6PPCh. 20 - Prob. 7PPCh. 20 - Prob. 8PPCh. 20 - Prob. 9PPCh. 20 - Practice Problem 20.11 In the preceding examples...
Ch. 20 - Prob. 11PPCh. 20 - Prob. 12PPCh. 20 - Practice Problem 20.14
Outline a synthesis of...Ch. 20 - Prob. 14PPCh. 20 - Prob. 15PPCh. 20 - PRACTICE PROBLEM
20.16
An amine A has the...Ch. 20 - PRACTICE PROBLEM Sulfonamides of primary amines...Ch. 20 - PRACTICE PROBLEM
20.18 (a) Starting with aniline...Ch. 20 - Prob. 19PCh. 20 - 20.20 Give common or systematic names for each of...Ch. 20 - Which is the most basic nitrogen in each compound?...Ch. 20 - Prob. 22PCh. 20 - Prob. 23PCh. 20 - Show how you might synthesize each of the...Ch. 20 - Prob. 25PCh. 20 - 20.26 Provide the major organic product from each...Ch. 20 - Prob. 27PCh. 20 - Prob. 28PCh. 20 - Prob. 29PCh. 20 - Prob. 30PCh. 20 - Prob. 31PCh. 20 - Write equations for simple chemical rests or state...Ch. 20 - Prob. 33PCh. 20 - 20.34 Using reactions that we have studied in this...Ch. 20 - 20.35 Provide a detailed mechanism for each of the...Ch. 20 - Prob. 36PCh. 20 - Prob. 37PCh. 20 - Prob. 38PCh. 20 - Prob. 39PCh. 20 - 20.40 Give structures for compounds R-W:
Ch. 20 - Prob. 41PCh. 20 - Prob. 42PCh. 20 - Diethylpropion (shown here) is a compound used in...Ch. 20 - Prob. 44PCh. 20 - 20.45 Compound W is soluble in dilute aqueous HCI...Ch. 20 - 20.46 Propose structures for compounds X, Y, and...Ch. 20 - Compound A(C10H15N) is soluble in dilute HCI. The...Ch. 20 - Prob. 48PCh. 20 - Prob. 49PCh. 20 - 20.52 When phenyl isochiocyanatc, , is reduced...Ch. 20 - Prob. 51PCh. 20 - 20.54 Propose a mechanism that can explain the...Ch. 20 - When acetone is treated with anhydrous ammonia in...Ch. 20 - Prob. 54PCh. 20 - Which of the following would be soluble in dilute...Ch. 20 - Which would yield propylamine? (d) Two of the...Ch. 20 - Select the reagent from the list below that could...Ch. 20 - Prob. 4QCh. 20 - 20.5 Select the stronger base from each pair (in...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
32. Compound X is insoluble in aqueous sodium bicarbonate. The IR spectrum of X has a strong absorption peak n...
Organic Chemistry
Determine the Coulombic and exchange energies for the following states, and determinewhich state is favored (ha...
Inorganic Chemistry
For fully developed laminar flow through a parallel-plate channel, the x-momentum equation has the form (d2udy2...
Fundamentals of Heat and Mass Transfer
Methyl nitrate, CH2NO2 , is used as a rocket propellant. The skeletal structure of the molecule is CH2ONO2. The...
General Chemistry: Principles and Modern Applications (11th Edition)
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (SYN) Draw the structure of the carboxylic acid that can be reacted with diazomethane to form each of the following compounds. (a) (b) (c) HN OCH3 %3arrow_forwardResveratrol is an antioxidant found in the skin of red grapes. Its anticancer, anti-inammatory, and various cardiovascular effects are under active investigation. (a) Draw all resonance structures for the radical that results from homolysis of the OH bond shown in red. (b) Explain why homolysis of this OH bond is preferred to homolysis of either OH bond in the other benzene ring.arrow_forwardDraw the structure of the following compounds all showing C and H atoms.(a) 2-methyl -3-iso propyl heptanes(b) Dicyclopropyl methane.arrow_forward
- 2. Question : Arrange the following compounds in increasing order of their reactivity in nucleophilic addition reactions (i) Ethanal, Propanal, Propanone, Butanone. (ii) Benzaldehyde, p-tolualdehyde, p-nitrobenzaldehyde, acetophenone.arrow_forward( provide detail explanation )arrow_forward! ( give step by step explanation)arrow_forward
- (a) Which of the following has lone pair on O atom that is part of aromaticity? (b) Which lacks complete cyclic conjugation? (c) Which does not follow the Huckel rule?arrow_forwardQuestion: Which of the following is an intermediate in the reaction of benzene with Br2 and AIBr3? * (b) (c) (d) Br H Br AIC Br H. H. H H H. H H. H H H. (A) Question: Which of the following can react readily either as a nucleophile or as an electrophile? * (CH3)3B (CH3)20 CH3CO2H H2C=CH2arrow_forwardRank the labeled H atoms in the following compound in order of increasing acidity. (See the attachment)arrow_forward
- (a) What functional group is undergoing a transformation in the reaction? (b) What functional group is it being transformed into (in the final product)?arrow_forwardA) Which compounds in the picture provided has an acidic group B) Which compounds in the picture provided has a basic group? Thank You!arrow_forwardDraw the structures of the following compounds. (Includes both new and old names.)(a) triphenylmethanolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY