
(a)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(a)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is phenyl formate.
The molecular formula of phenyl formate is
The structure of phenyl formate is given as,
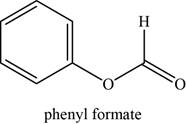
Figure 1
(b)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(b)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is cyclohexyl benzoate.
The molecular formula of cyclohexyl benzoate is
The structure of cyclohexyl benzoate is given as,
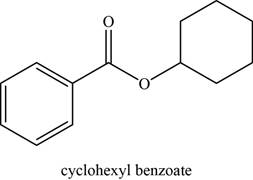
Figure 2
(c)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(c)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is cyclopentyl phenylacetate.
The molecular formula of cyclopentyl phenylacetate is
The structure of cyclopentyl phenylacetate is given as,
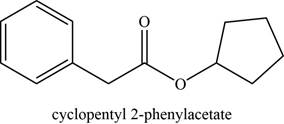
Figure 3
(d)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(d)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is N-butylacetamide.
The molecular formula of N-butylacetamide is
The structure of N-butylacetamide is given as,
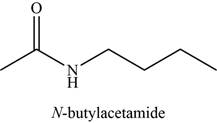
Figure 4
(e)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(e)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is N,N-dimethylformamide.
The molecular formula of N,N-dimethylformamide is
The structure of N,N-dimethylformamide is given as,
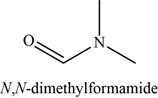
Figure 5
(f)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(f)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is benzoic propionic anhydride.
The molecular formula of benzoic propionic anhydride is
The structure of benzoic propionic anhydride is given as,
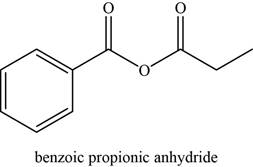
Figure 6
(g)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(g)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is benzamide.
The molecular formula of benzamide is
The structure of benzamide is given as,
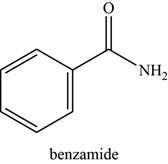
Figure 7
(h)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(h)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is
The molecular formula of
The structure of
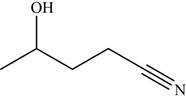
Figure 8
(i)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(i)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is
The molecular formula of
The structure of
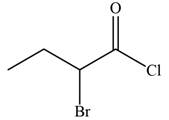
Figure 9
(j)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(j)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is
The molecular formula of
The structure of
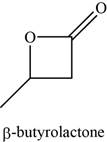
Figure 10
(k)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(k)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is phenyl isocyanate.
The molecular formula of phenyl isocyanate is
The structure of phenyl isocyanate is given as,
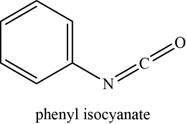
Figure 11
(l)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(l)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is cyclobutyl ethyl carbonate.
The molecular formula of cyclobutyl ethyl carbonate is
The structure of cyclobutyl ethyl carbonate is given as,
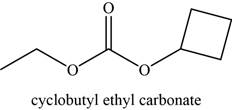
Figure 12
(m)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(m)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is
The molecular formula of
The structure of
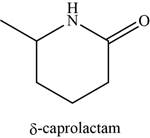
Figure 13
(n)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(n)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is trichloroacetic anhydride.
The molecular formula of trichloroacetic anhydride is
The structure of trichloroacetic anhydride is given as,
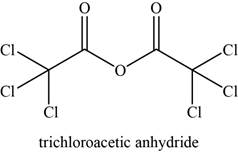
Figure 14
(o)
Interpretation:
The structure to correspond with the given common and systematic names is to be drawn.
Concept introduction:
Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because common names do not follow any rule, whereas IUPAC names follow specific rules. The common name does not include any suffix, prefix, and numbers.
(o)
Answer to Problem 21.42SP
The structure to correspond with the given common and systematic names is given below.
Explanation of Solution
The given compound is Ethyl-N-methyl carbamate.
The molecular formula of Ethyl-N-methyl carbamate is
The structure of Ethyl-N-methyl carbamate is given as,
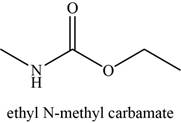
Figure 15
Want to see more full solutions like this?
Chapter 21 Solutions
ORGANIC CHEMISTRY
- QUESTION 1 Which of the following compounds has an activating group? A. Benzoic acid B. None in the answer C. Bromobenzene D. Diphenylamine QUESTION 2 Which of the following compounds has a deactivating group attached to the benzene ring? A. Toluene B. Acetanilide C. Phenol D. Salicyclic Acidarrow_forwardReduction of an alkyl azide results in the formation of —-. A. an imine B. an oxime C. a tertiary amine D. a secondary amine E. a primary aminearrow_forward1. An alkene reacts with water with an acid catalyst results into a formation of: A. Aldehyde B. Ketone C. Alcohol D. Ester 2. 3-Methylhexanal with K2Cr2O7 will yield: A. 3-Methyl-1-hexanol B. 3-Methylhexanoic acid C. 3-Methyl-1-hexanone D. 3-Methyl-1-hexanethiol 3. This is a reverse process of Hydration reaction: A. Oxidation reaction B. Reduction reaction C. Dehydration reaction D. Hydration reaction 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forward
- 1. A pleasant smelling liquid having a boiling point of 101°C. What is the structure of this compound? Major IR Absorptions Major lons in the MS 2880-2980 cm-¹ 29 41 1737 (str) 1194 (str) 1166 (str) 56 57 (base) 73 85 101 (small) 116 (molecular ion) 100 1737 500 vavenumber cm-1 157 Transmittance %T ģ 0. liquid film sample 100 80 60 4000 Relative Intensity 9 20 0 10 20 2977 9000 2876 30 4. 40 2000 50 56 60 m/z 70 1500 73 80 1194 85 1166 90 1000 101 repla 100 110 M: 116arrow_forwardAmong which of the following pairs can be differentiated by Iodoform Test? A. Benzaldehyde and formaldehyde B. Acetaldehyde and acetone C. Benzaldehyde and 3-pentanone D. Formaldehyde and acetaldehyde Explain your answerarrow_forwardDraw a structure for each of the following: a. N,N-dimethylhexanamide b. 3,3-dimethylhexanamide c. cyclohexanecarbonyl chloride d. propanenitrilee. propionamide f. sodium acetate g. benzoic anhydride h. beta-valerolactone i. 3-methylbutanenitrile j. cycloheptanecarboxylic acid k. benzoyl chloridearrow_forward
- 10. Under certain conditions interacts with sodium nitrite: A. Resorcinol B. Nicotinic acid C. Butadion D. Morphine hydrochloride E. Benzoic acidarrow_forwardCyclopentanone is treated with chlorine (Cl2) in the presence of acid (H*). What product is formed? A. Chlorocyclopentane B. 1-chloro-1-hydroxycyclopentane C. 2-chlorocyclopentanone D. 3-chlorocyclopentanonearrow_forwardWhat compounds are formed from the reaction of benzoyl chloride with the following reagents? a. sodium acetate b. water c. excess dimethylamine d. aqueous HCl e. aqueous NaOH f. cyclohexanol g. excess benzylamine h. 4-chlorophenol i. isopropyl alcohol j. excess aniline k. potassium formatearrow_forward
- Consider the following compounds: A. 2-Pentenal B. 2-Heptanol C. Hexanamide D. Propionyl Chloride Е. 3-Нехanone F. y-Phenylbutyric Acid G. Ethyl Chloroacetate Н. 1-Неptanol I. Acetic Anhydride What simple chemical test can be done to tell the difference between (choose only one correct answer from the choices below, only put in the capital letter in the boxes, there could be more than one correct answer): Between Compound C and Compound D, you can use the following Between Compound A and Compound G, you can use the following Between Compound B and Compound I, you can use the following Between Compound E and Compound F, you can use the following Between Compound B and Compound H, you can use the following A. Add HCI (ag) solution to both. B. Add Tollen's reagent to both. C. Add KOI reagent to both. D. Add Br,/CCI, to both. E. Add NaHCO3 (aq) solution to both. F. Add Acetyl Chloride to both. G. Add HCRO4/A to both. H. Beilstein Flame Test on both. I. Ferrous Hydroxide Test on both. J.…arrow_forward2. Draw out the following compounds. a. N-methylaniline b. Triisopropylamine c. N,N-dipropylhexylamine d. 1,5-pentanediamine (Also known as Cadaverine for its smell)arrow_forwardHelparrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

