
(a)
Interpretation:
The reagent has to be proposed for the conversion of A to B.
Concept introduction:
Michael reaction:
The nucleophile is reaction with α,β-unsaturated carbonyl compound (1,4 –addtion) which yield the addition product is called Michael reaction.
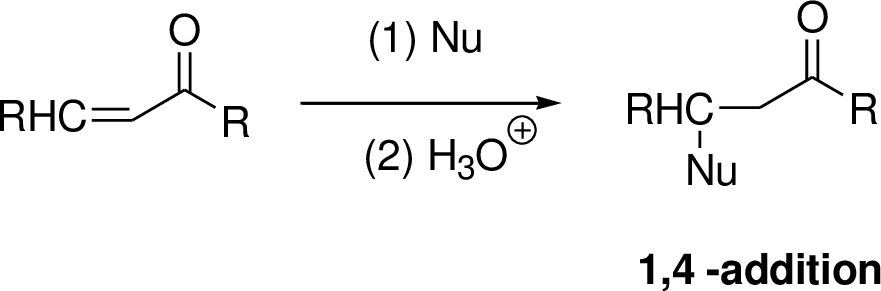
(b)
Interpretation:
The reagent has to be proposed for the conversion of B to C.
Concept introduction:
Reduction:
(c)
Interpretation:
The reagent has to be proposed for the conversion of C to D.
Concept introduction:
Chlorination: Alcohols reaction with thionyl chloride gives chlorinated compound.
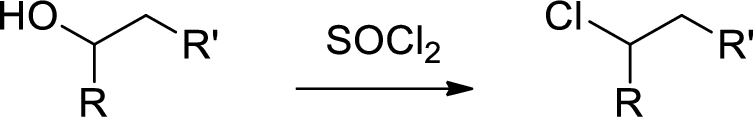
(d)
Interpretation:
The mechanism has to be proposed for the conversion of E to F and the formation of methyl chloride has to be shown through mechanism.
(e)
Interpretation:
The reagent has to be proposed for the conversion of F to Fluoxetine.
Concept introduction:
Hydrolysis: Amide undergoes hydrolysis using base like sodium hydroxide gives acid.
(f)
Interpretation:
The possible stereoisomer’s has to be shown if the product is chiral.
Concept introduction:
Chiral:
A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.arrow_forwardShow how the following ketones might be synthesized from the indicated acids, usingany necessary reagents.(b) methyl cyclohexyl ketone from cyclohexanecarboxylic acidarrow_forwardIdentify products A and B from the given 1H NMR data. Treatment of acetone [(CH3)2C=O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1H) ppm. What is the structure of B?arrow_forward
- The following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardAnswer ALL parts. (a) Provide reagents for the following transformation. No mechanism is required. NH2 (b) Suggest structures for A and B, and provide detailed mechanisms for their formation. 1. CI он NaOH (2eq) A ACONA/ACOH B 2. H3O* heat (c) Provide a detailed mechanism for the following transformation and suggest a chemical structure for C. All reagents are added together and no additional acid or base is needed. MENH2 COME C9H13NO2 lo (d) Suggest a retrosynthetic analysis for the following molecule, and outline a synthetic route, specifying reagents. No mechanisms are required. 3.arrow_forwardWhat reagents would you use to carry out the following reaction? (A) H₂ / Pt (B) (C) (D) H₂/Lindlar's ??? Li, EtNH₂ followed by NH4Cl H₂O₂ / NaOHarrow_forward
- Compound A undergoes an acid-catalyzed hydrolysis. One of the products (B) that is isolated gives the following 1H NMR spectrum. Identify the compounds A and Carrow_forwardA chemist was trying to synthesize compound C over the course of two steps, butafter the first step they realized that they had two different products (A and B). Both A and B had peaks that corresponded to a ketone and an ester( A has aketone and an ester, and B has a ketone and an ester). Provide the structure for A and Barrow_forward(a) 4 Choose the major product of the following elimination. (b) OH H₂SO4 heat (c) (d) 1 ptsarrow_forward
- When phenyl 3,4-dimethylbenzoate undergoes electrophilic aromatic substitution, one ring reacts much more readily than the other. (a) Which one is it? Encircle the ring (b) Explain your answer. (c) Draw the resulting product/s when it undergoes bromination reaction. Product (c) (a) Explanation (b)arrow_forwardDraw a structural formula for the product formed by treating butanal with each reagent. (a) LiA1H4LiA1H4 followed by H2OH2O (b) NaBH4NaBH4 in CH3OH/H2O (c) H2/Pt (d) Ag(NH3)2+in NH3/H2O (e) H2CrO4, heat (f) HOCH2CH2OH,HClarrow_forwardHow will you synthesize cyclohexanecarboxaldehyde (cyclohexylmethanal) from the following reagents? (There are no restrictions on the reagents or the number of steps). (a) Cyclohexanone (b) Ethynylcyclohexane (c) Methyl cyclohexylformate (Remember: Formic acid is the IUPAC recognized name for Methanoic acid) (d) Cyclohexanecarboxylic acid (Cyclohexylmethanoic acid) (e) Vinylcyclohexanearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
