
Bundle: Physical Chemistry, 2nd + Student Solutions Manual
2nd Edition
ISBN: 9781285257594
Author: David W. Ball
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 21.5E
Figure
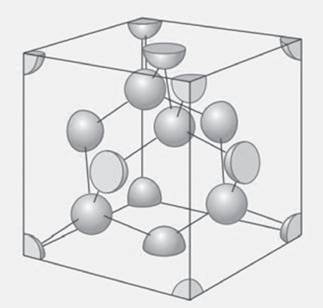
Figure 21.35 The unit cell of diamond. See exercise
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The accompanying figure shows three different crystallographic planes for a unit cell of a hypothetical metal. The circles represent atoms. Draw its unit cell and show the atoms’ positions. To what crystal system does the unit cell belong and what would this crystal structure be called?
Based on the atom arrangement/layering and unit cell packing, what is the cell type of CsCl?
a. simple cubic
b. body-centered cubic
c. face-centered cubic
*Refer to the image below for the crystal structure.
*Please explain thoroughly.
1. Given the following data, calculate the lattice energy of aluminum oxide, Al2O3.
a.-13 272 kJ mol-1b.-8300 kJ mol-1c.-15 333 kJ mol-1d. -10 610 kJ mol-12. A face-centered cubic unit cell is the repeating unit in which type of crystal packing?
a.simple
b.cubic close-packed
c.hexagonal close-packed
d. body-centered
Chapter 21 Solutions
Bundle: Physical Chemistry, 2nd + Student Solutions Manual
Ch. 21 - Prob. 21.1ECh. 21 - Boron nitride, BN, is a very hard material, harder...Ch. 21 - Prob. 21.3ECh. 21 - Prob. 21.4ECh. 21 - Figure 21.35 shows a unit cell of diamond....Ch. 21 - Prob. 21.6ECh. 21 - How many different unit cells can a crystal have...Ch. 21 - Prob. 21.8ECh. 21 - Prob. 21.9ECh. 21 - Prob. 21.10E
Ch. 21 - Prob. 21.11ECh. 21 - Prob. 21.12ECh. 21 - Prob. 21.13ECh. 21 - Prob. 21.14ECh. 21 - Prob. 21.15ECh. 21 - Prob. 21.16ECh. 21 - Prob. 21.17ECh. 21 - Prob. 21.18ECh. 21 - Prob. 21.19ECh. 21 - Prob. 21.20ECh. 21 - Prob. 21.21ECh. 21 - Prob. 21.22ECh. 21 - Prob. 21.23ECh. 21 - Prob. 21.24ECh. 21 - Prob. 21.25ECh. 21 - Prob. 21.26ECh. 21 - Prob. 21.27ECh. 21 - Prob. 21.28ECh. 21 - For a simple cubic lattice, what is the ratio of...Ch. 21 - Prob. 21.30ECh. 21 - Prob. 21.31ECh. 21 - Consider Figure 21.21. If the lower rightmost...Ch. 21 - Prob. 21.33ECh. 21 - The aluminum-nickel alloy AlNi has a simple cubic...Ch. 21 - Prob. 21.35ECh. 21 - The first two signals from a powdered sample has X...Ch. 21 - Prob. 21.37ECh. 21 - Prob. 21.38ECh. 21 - Prob. 21.39ECh. 21 - Prob. 21.40ECh. 21 - Prob. 21.41ECh. 21 - Prob. 21.42ECh. 21 - Prob. 21.43ECh. 21 - Prob. 21.44ECh. 21 - Prob. 21.45ECh. 21 - What is the coordination number in the cesium...Ch. 21 - Prob. 21.47ECh. 21 - Which solid phase that is, which allotrope of...Ch. 21 - Prob. 21.49ECh. 21 - Prob. 21.50ECh. 21 - Write Born-Haber cycles showing the relationship...Ch. 21 - Prob. 21.52ECh. 21 - Prob. 21.53ECh. 21 - Prob. 21.54ECh. 21 - The lattice energy for potassium iodide, KI, is...Ch. 21 - Prob. 21.56ECh. 21 - Prob. 21.57ECh. 21 - Prob. 21.58ECh. 21 - Prob. 21.59ECh. 21 - Prob. 21.60ECh. 21 - Prob. 21.61ECh. 21 - Prob. 21.62ECh. 21 - Prob. 21.63ECh. 21 - Prob. 21.64E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Phase diagrams for materials that have allotropes can be more complicated than those shown in the chapter. Use the phase diagram for carbon given here to answer the following questions. (a) How many triple points are present and what phases are in equilibrium for each? (b) Is there a single point where all four phases are in equilibrium? (c) Which is more stable at high pressures, diamond or graphite? (d) Which is the stable phase of carbon at room temperature and 1 atmosphere pressure?arrow_forwardAn amorphous solid can sometimes be converted to a crystalline solid by a process called annealing. Annealing consists of heating the substance to a temperature just below the melting point of the crystalline form and then cooling it slowly. Explain why this process helps produce a crystalline solid.arrow_forward1. A metallic solid ingot has a mass of 14.7 grams. It was loaded into a graduated cylinder containing 40.0 mL or water. Upon addition of the ingot, the volume of the water and the ingot was 41.6 mL. What is the density of the ingot? 2. Assuming the solid ingot in question 1 is polonium (Po, element 84) which packs in a primitive cubic unit cell structure calculate the packing efficiency of the unit cell? Atoms touch along edge. a = 2r Carrow_forward
- hypothetical metal has an orthorhombic unit cell for which the a, b, and c lattice parameters are 0.675, 0.532, and 0.897 nm, respectively. If the atomic packing factor, atomic radius and atomic weight are 0.732, 0.225 nm and 115.80 g/ mol, respectively. compute its density. a. 2.58 g/nm3 b. 7.68 g/nm3 c. 4.96 g/nm3 d. 8.50 g/nm3 е. not all the above f. 1.84 g/nm3 g. 4.77 g/nm3 h. 1.23 g/nm3arrow_forwardIndium has a tetragonal unit cell for which the a and c lattice parameters are 0.459 and 0.495nm, respectively. a. If the atomic packing factor and atomic radius are 0.693 and 0.1625 nm, respectively, determine the number of atoms in each unit cell. b. The atomic weight of indium is 114.82 g/mol; compute its theoretical density.arrow_forwardDefine unit cellarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=HCWwRh5CXYU;License: Standard YouTube License, CC-BY