
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 27P
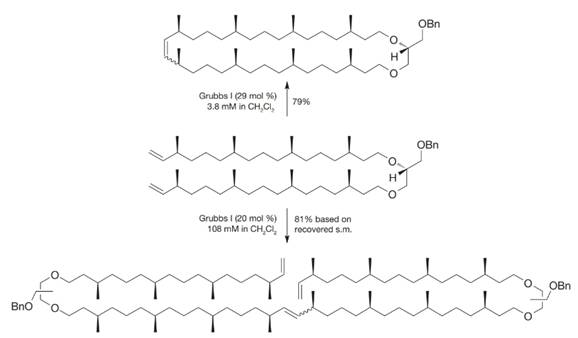
When the following molecule was exposed to the Grubbs I initiator at different concentrations, different products resulted. How can you account for these different outcomes?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the best way to turn benzene into the following product? Would friedel-crafts alkylation followed by some sort of nitration work or would that be too low yield?
Complete the reaction schemes below providing the reagents required to achieve thetransformation. More than one step may be necessary for each scheme.
I'm having trouble proposing a syntheses for this molecule using the reatant given. Can you please help show how to approach this problem?
Chapter 21 Solutions
Organic Chemistry
Ch. 21 - PRACTICE PROBLEM 21.1
For each of the following...Ch. 21 - Prob. 2PPCh. 21 - PRACTICE PROBLEM 21.3 What product would you...Ch. 21 - Prob. 4PPCh. 21 - PRACTICE PROBLEM 21.5 What is the product of the...Ch. 21 - Prob. 6PPCh. 21 - Prob. 7PPCh. 21 - Prob. 8PPCh. 21 - Prob. 9PPCh. 21 - Prob. 10PP
Ch. 21 - Prob. 11PPCh. 21 - Practice Problem 21.12 What products would form...Ch. 21 - Prob. 13PCh. 21 - Prob. 14PCh. 21 - How would you synthesize each of the following...Ch. 21 - Prob. 16PCh. 21 - Predict the product(s) for each of the following...Ch. 21 - Prob. 18PCh. 21 - Prob. 19PCh. 21 - Prob. 20PCh. 21 - Prob. 21PCh. 21 - 21.22 Write a mechanism that can account for the...Ch. 21 - Prob. 23PCh. 21 - Prob. 24PCh. 21 - Prob. 25PCh. 21 - 21.26 In 1985, T. Katz (Columbia University)...Ch. 21 - When the following molecule was exposed to the...Ch. 21 - During the course of the following Stille...Ch. 21 - 1. In “The Chemistry of... Complex Cross...Ch. 21 - Prob. 2LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Exercise 2
By using a conversion factor from the back inside cover, determine the length in kilometer...
Chemistry: The Central Science (14th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
2.81 In which of the fo1losing pairs do both numbers contain the same number of significant figures? (2.2)
a....
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
21.49 The atomic masses of hydrogen-2 (deuterium), helium-4, and lithium-6 are 2.014102 amu, 4.002602 amu, 6.01...
Chemistry: The Central Science (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide the necessary sequence of reactions to generate the products below from their respective starting materials.arrow_forwardMaking use of the corresponding mechanisms and additional comments, justify the following observations A) Mechanism of Formation of the super-electrophile B) COMPLETE mechanism meta position C) COMPLETE mechanism para position D) Explanation of the resultarrow_forwardHow to get the molecule given below by getting out of bromobenzene and using the reagents you want? You synthesize? Show it.arrow_forward
- In the chemical reaction between crotyl chloride and ethanolic silver nitrate solution, a precipitate is definitely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.arrow_forwardPredict the coupling products of the organocuprate, Heck, and Suzuki reactions,and use these reactions in synthesesarrow_forwardPropose a mechanism and use stability factors to account for the formation of a major product in the reaction of pyrrole with Br2, FeBr3.arrow_forward
- After E2 elimination, will the following molecule contain deuterium? Why or why not?arrow_forwardQuestion: How do quantum mechanical effects influence the stability and reactivity of molecules with non-classical carbocations, such as the 2-norbornyl cation, and how does this impact the reaction mechanisms and outcomes?arrow_forwardthe following reactions will not produce the product given. briefly explain why.arrow_forward
- In an aqueous solution containing sodium bicarbonate, aniline reacts quickly with bromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions, however, and the yields (mostly m-nitroaniline) are poor.Explain why nitration of aniline is so sluggish and why it gives mostly metasubstitution.arrow_forwardPredict the major products of treating the following compounds with hot, concentratedpotassium permanganate, followed by acidification with dilute HCl. (tetralin)arrow_forwardShow how to accomplish the following synthesis in a good yield.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY