
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 26P
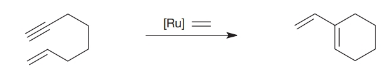
In 1985, T. Katz (Columbia University) discovered that
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Complete the following by drawing products for a and b, and also identify the diene and dienophile in each one.
a. Propose a mechanism for the following reaction. (Hint: An electrocyclic reaction is followed by a Diels–Alder reaction.)b. What would be the product if trans-2-butene were used instead of ethene?
Diels Adler reaction-- predict the product--- provide mechanism of formation.
Chapter 21 Solutions
Organic Chemistry
Ch. 21 - PRACTICE PROBLEM 21.1
For each of the following...Ch. 21 - Prob. 2PPCh. 21 - PRACTICE PROBLEM 21.3 What product would you...Ch. 21 - Prob. 4PPCh. 21 - PRACTICE PROBLEM 21.5 What is the product of the...Ch. 21 - Prob. 6PPCh. 21 - Prob. 7PPCh. 21 - Prob. 8PPCh. 21 - Prob. 9PPCh. 21 - Prob. 10PP
Ch. 21 - Prob. 11PPCh. 21 - Practice Problem 21.12 What products would form...Ch. 21 - Prob. 13PCh. 21 - Prob. 14PCh. 21 - How would you synthesize each of the following...Ch. 21 - Prob. 16PCh. 21 - Predict the product(s) for each of the following...Ch. 21 - Prob. 18PCh. 21 - Prob. 19PCh. 21 - Prob. 20PCh. 21 - Prob. 21PCh. 21 - 21.22 Write a mechanism that can account for the...Ch. 21 - Prob. 23PCh. 21 - Prob. 24PCh. 21 - Prob. 25PCh. 21 - 21.26 In 1985, T. Katz (Columbia University)...Ch. 21 - When the following molecule was exposed to the...Ch. 21 - During the course of the following Stille...Ch. 21 - 1. In “The Chemistry of... Complex Cross...Ch. 21 - Prob. 2LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
a. Prepare a molecular orbital energy-level diagram for NO showing clearly how the atomic orbitals interact to ...
Inorganic Chemistry
Referring to the two ligands, A and B, described in Problem 21.80, which complex would be expected to be more e...
Chemistry: The Molecular Nature of Matter
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
In qualitative analysis, Ca2+ and Ba2+ are separated from Na+, K+, and Mg2+ by adding aqueous (NH4)2CO3 to a so...
General Chemistry: Atoms First
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The aryl diene undergoes sequential Heck reactions to give a product with the molecular formula C15H18. Propose a structural formula for this product.arrow_forwardSuppose you were told that each reaction is a substitution reaction, but you were not told the mechanism. Describe how you could conclude from the structure of the haloalkane or cycloalkene, the nucleophile, and the solvent that each reaction is an SN1 reaction.arrow_forwardExplain, with reference to the mechanism, why addition of one equivalent of HCl to diene A forms only two products of electrophilic addition, even though four constitutional isomers are possible.arrow_forward
- Friedel–Crafts alkylations can be carried out with carbocations formed from reactions other than the reaction of an alkyl halide with AlCl3. Propose a mechanism for the following reaction:arrow_forwardpropose a retrosynthesis, and a way in which this diene can be synthesizedarrow_forwarda) hydrogen bromide with sodium methoxide base from trans-1-bromo 2-methyl cyclohexane the product that will be formed by considering the conformation of the starting compound when it is removed Find the mechanism of the reaction by writing and explaining. b) Write down the mechanism of the reaction of methylcyclohexene with bromine under UV light. Find the resulting product.arrow_forward
- From hofmann elimination,propose the major product and demonstrate the mechanism to obtain the major product for each reactionarrow_forwardWhat is the major substitution product for the following reaction? Show the mechanism for the reaction.arrow_forwardPropose a mechanism for this reaction. Account for both its regioselectivity and stereoselectivity.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY