
Interpretation:
The other functional group is identified in Capsaicin other than phenol.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Amide:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with nitrogen is called as amide.
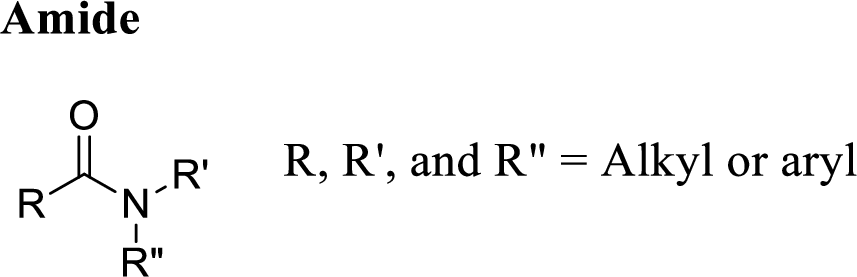
Alkenes are a class of hydrocarbons. The carbon-carbon double bond is called as alkenes and it is also called as olefins.
Ether:
The aliphatic or
Example is given below
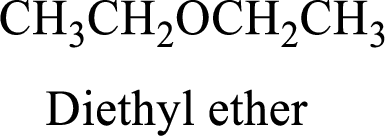
Trending nowThis is a popular solution!

Chapter 21 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- 2. Oxybenzone and 2-ethylhexyl-p-methoxycinnamate are UV absorbing compounds used in sunscreens. Identify and encircle the functional groups in these compounds. a. Oxybenzone Ö HÓ b. 2-ethylhexyl-p-methoxycinnamatearrow_forwardWhat are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. acetic acid and toluene b. phenylic acid and ethanol C. ethanoic acid and benzene d. benzoic acid and ethanol e. phenylic acid and methanol O a O barrow_forwardamine, (2) an amide, or (3) both an amine and an amide. 17-106 Classify each of the following compounds as (1) an amine, (2) an amide, or (3) both an amine and an NH2 b. `NH a. H2N H d. с.arrow_forward
- 14. PART 3: Draw the structure for compound A.arrow_forward4. Tylenol also is an analgesic often taken by people allergic to aspirin. The active ingredient is acetaminophen. HO- -NH H3C Would acetaminophen give a positive phenol test? (Y/N). Does this compound contain the ester functional group? If not, what functional group(s) is/are present?arrow_forwardSelect all functional groups present in the following structure of a drug commonly found in a dentist's office N a) Alkyl halide b) Alcohol c) Carboxylic acid d) Phenol e) Amine f) Ether Og) Amide h) Ester i) Ketone 0 j) Aromatic ring (aka phenyl) (aka arene)arrow_forward
- 16. An atom or group of atoms that can give organic compounds distinct chemical and physical properties. 21. When a compound with the general formula R-COOH loses a proton, the product that remains is described with this term. Its general formula is R-COO- 24. A class of organic compounds in which three or more carbons form a ring structure. All of the carbon-to-carbon bonds are single bonds in this family of compounds.arrow_forward2. What is produced when an amine reacts with water? A. A primary alcohol and ammonia B. An amide and a hydrogen (H+) ion C. An ammonium ion and a hydroxide (OH-) ion D. An amide and a hydroxide (OH-) ionarrow_forward4. The organic starting materials for the preparation of an ester could be C. a ketone and alcohol A an acid and alcohol B. water and oxygen D. alkane and aldehydearrow_forward
- 2. a. What does this product smell like? b. What natural product does it come from?arrow_forwardA lot of controversy surrounds the use of Aspartame as an artificial sweetener. The main argument is concerned with the production of methanol in the body. From the structure of aspartame given below: G0₂CH3 HẠN CH—ỆNH—CHCH, CH, COCH Source for the formation of methanol is, A. ester group in the compound B. amide group in the compound C. carboxylic acid group in the compound D. amino group in the compound E. benzene ring in the compoundarrow_forward1. Amines are classified according to the number of alkyl or aryl groups directly attached to a certain atom in the molecule. What atom is this? 2. Draw examples of a primary, secondary, and tertiary amine. Secondary Primary H N-CH3 Tertiary CHS N-CH3 CH CHa N-CHS 3. What are the names of CH3CH2-NH, in the IUPAC and common naming systems? 4. Complete the following equation and name the organic reactants and products. CH,NH, (aq) + H,O = 5. What is the general name of the product of the neutralization reaction between an alkylamine and an acid? AIKVI Ammonium Salt laarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



