
ORGANIC CHEMISTRY-EBOOK>I<
9th Edition
ISBN: 9781305084414
Author: McMurry
Publisher: INTER CENG
expand_more
expand_more
format_list_bulleted
Question
Chapter 21.SE, Problem 60AP
Interpretation Introduction
Interpretation:
The step-growth
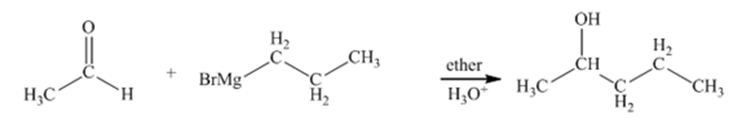
Concept introduction:
This step growth
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The reaction of p-cresol with CH2=O resembles the reaction of phenol (PhOH) with CH2=O, except that the resulting polymer is thermoplastic but not thermosetting. Draw the structure of the polymer formed, and explain why the properties of these two polymers are so different.
Polystyrene is a synthetic aromatic hydrocarbon polymer made from the
monomer known as styrene.
H
H
H
Polystyrene
(i)
Draw the structure of styrene.
Outline the mechanism when styrene reacts with HCl, following
(ii)
Markovnikov's rule.
The reaction of p-cresol with CH2=O resembles the reaction of phenol (PHOH) with CH2 = 0, except that the resulting polymer
is thermoplastic but not thermosetting. Draw the structure of the polymer formed, and explain why the properties of these two
polymers are so different.
CH3
OH
+ CH2=0
p-cresol
Chapter 21 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
Ch. 21.1 - Give IUPAC names for the following substances:Ch. 21.1 - Draw structures corresponding to the following...Ch. 21.2 - Prob. 3PCh. 21.2 - Rank the compounds in each of the following sets...Ch. 21.2 - Predict the products of the following nucleophilic...Ch. 21.2 - Prob. 6PCh. 21.3 - Prob. 7PCh. 21.3 - If the following molecule is treated with acid...Ch. 21.4 - How might you prepare the following esters using a...Ch. 21.4 - Prob. 10P
Ch. 21.4 - Prob. 11PCh. 21.4 - Prob. 12PCh. 21.4 - Prob. 13PCh. 21.5 - Prob. 14PCh. 21.5 - What product would you expect from reaction of one...Ch. 21.6 - Prob. 16PCh. 21.6 - Prob. 17PCh. 21.6 - Show the products you would obtain by reduction of...Ch. 21.6 - What ester and what Grignard reagent might you...Ch. 21.7 - Prob. 20PCh. 21.7 - How would you use the reaction of an amide with...Ch. 21.8 - Write the mechanism of the reaction shown in...Ch. 21.9 - Prob. 23PCh. 21.9 - Prob. 24PCh. 21.10 - Prob. 25PCh. 21.10 - Prob. 26PCh. 21.SE - Name the following compounds:Ch. 21.SE - Prob. 28VCCh. 21.SE - Prob. 29VCCh. 21.SE - Prob. 30VCCh. 21.SE - Predict the product(s) and provide the mechanism...Ch. 21.SE - Predict the product(s) and provide the mechanism...Ch. 21.SE - Predict the product(s) and provide the mechanism...Ch. 21.SE - Predict the product(s) and provide the complete...Ch. 21.SE - Prob. 35MPCh. 21.SE - When 4-dimethylaminopyridine (DMAP) is added in...Ch. 21.SE - Prob. 37MPCh. 21.SE - Prob. 38MPCh. 21.SE - Prob. 39MPCh. 21.SE - The hydrolysis of a biological thioester to the...Ch. 21.SE - Prob. 41MPCh. 21.SE - Prob. 42MPCh. 21.SE - Prob. 43MPCh. 21.SE - In the iodoform reaction, a triiodomethyl ketone...Ch. 21.SE - Give IUPAC names for the following compounds:Ch. 21.SE - Prob. 46APCh. 21.SE - Draw and name compounds that meet the following...Ch. 21.SE - Predict the product, if any, of reaction between...Ch. 21.SE - Prob. 49APCh. 21.SE - Prob. 50APCh. 21.SE - What product would you expect to obtain from...Ch. 21.SE - Prob. 52APCh. 21.SE - Prob. 53APCh. 21.SE - The following reactivity order has been found for...Ch. 21.SE - Prob. 55APCh. 21.SE - Outline methods for the preparation of...Ch. 21.SE - Prob. 57APCh. 21.SE - When ethyl benzoate is heated in methanol...Ch. 21.SE - tert-Butoxycarbonyl azide, a reagent used in...Ch. 21.SE - Prob. 60APCh. 21.SE - Prob. 61APCh. 21.SE - What is the structure of the polymer produced by...Ch. 21.SE - Polyimides with the structure shown are used as...Ch. 21.SE - Prob. 64APCh. 21.SE - Propose a structure for a compound, C4H7ClO2, that...Ch. 21.SE - Assign structures to compounds with the following...Ch. 21.SE - Prob. 67APCh. 21.SE - When a carboxylic acid is dissolved in...Ch. 21.SE - Prob. 69APCh. 21.SE - Prob. 70APCh. 21.SE - Prob. 71APCh. 21.SE - Phenyl 4-aminosalicylate is a drug used in the...Ch. 21.SE - N,N-Diethyl-m-toluamide (DEET) is the active...Ch. 21.SE - Tranexamic acid, a drug useful against blood...Ch. 21.SE - One frequently used method for preparing methyl...Ch. 21.SE - Prob. 76APCh. 21.SE - Assign structures to compounds with the following...Ch. 21.SE - Propose structures for compounds with the...Ch. 21.SE - Propose a structure for the compound with the...Ch. 21.SE - Draw the structure of the compound that produced...Ch. 21.SE - Prob. 81APCh. 21.SE - Epoxy adhesives are prepared in two steps. SN2...
Knowledge Booster
Similar questions
- Polystyrene is produced commercially by reaction of styrene with butyllithium as an anionic initiator. Using resonance structures, explain how the chain-carrying intermediate is stabilizedarrow_forwardEpoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichlorohydrin forms a prepolymer, which is then cured by treatment with a triamine such as H2NCH2CH2NHCH2CH2NH2. Draw structures to show how addition of the triamine results in a strengthening of the polymer.arrow_forwardPolycarbonates (Section 29.5C) are also formed by using a nucleophilic aromatic substitution route (Section 22.3B) involving aromatic difluoro monomers and carbonate ion. Propose a mechanism for this reaction.arrow_forward
- Draw a structural formula of the polymer resulting from base-catalyzed polymerization of each compound. Would you expect the polymers to be optically active? (S)-(+)-lactide is the dilactone formed from two molecules of (S)-(+)-lactic acid.arrow_forwardDraw a structural formula for the polymer formed in the following reactionsarrow_forwardCarototoxin is a natural pesticide produced by carrots, with formula C₁H2O. It undergoes hydrogenation with Pd to: give product A, C,H,O, and with Lindlar's catalyst to give product B, C,H2O. Ozonolysis followed by zinc leads to a mixture of methanal, octanal, 1,2-ethanedioic acid, 3-oxopropanoic acid, and 2-hydroxy-3-oxopropanoic acid. Draw possible structures for carototoxin, A, and B. Product B structure H2, Lindlar's Structure of carototoxin H2, Pd/C Product A structurearrow_forward
- Explain the effects the addition of glycerol (a triol) will have on the molecular architecture of the following polymerization reactions: a) Condensation polymerization of terephthalic acid + ethylene glycol b) Condensation polymerization of 6-hydroxyhexanoic acidarrow_forwardBakelite, a durable plastic used for the casing of early radios and TVs, was the first of the thermosetting polymers. It is a highly cross-linked polymer formed from the acid-catalyzed polymerization of phenol and formaldehyde. Because it is much darker than Melmac, Bakelite’s range of colors is limited. Propose a structure for Bakelite.arrow_forwardSharpless epoxidation of allylic alcohol X forms compound Y. Treatment of Y with NaOH and C6H5SH in an alcohol–water mixture forms Z. Identify the structure of Y and draw a mechanism for the conversion of Y to Z. Account for the stereochemistry of the stereogenic centers in Z. Z has been used as an intermediate in the synthesis of chiral carbohydrates.arrow_forward
- Tritan is a polymer marketed to consumers looking for BPA-free products. Although the detailed structure of Tritan is protected by patent, it is known to be a polyester (not a polycarbonate) composed of three monomers—dimethyl terephthalate, 2,2,4,4- tetramethylcyclobutane-1,3-diol, and 1,4-cyclohexanedimethanol. Propose a possible structure for Tritan from dimethyl terephthalate and the two diols drawn.arrow_forwardCaprolactam, the monomer from which nylon 6 is synthesized, is prepared from cyclo- hexanone in two steps. In Step 1, cyclohexanone is treated with hydroxylamine to form cyclohexanone oxime. Treatment of the oxime with concentrated sulfuric acid in Step 2 gives caprolactam by a reaction called a Beckmann rearrangement. Propose a mecha- nism for the conversion of cyclohexanone oxime to caprolactam. HO NH NH,OH H,SO, Cyclohexanone Cyclohexanone Сaprolactam oximearrow_forwardThermosetting resins similar to Bakelite (Section 31.7) have also been prepared from renewable feedstocks. One method uses cardinol, the major constituent of the liquid obtained from roasted cashew nutshells. What polymer is obtained when cardinol is treated with formaldehyde (H2C=O) in the presence of a proton source? он H2C=0 H+ cardinolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole