
Concept explainers
(a)
Interpretation:
The more acidic group −NH3+ or −COOH in the neutral amino acid alanine needs to be determined.
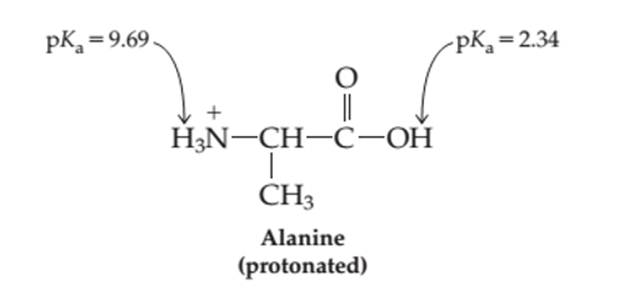
Concept introduction:
Proteins are
(b)
Interpretation:
The percentage of each form-protonates (H2A+ ), neutral (HA) and deprotonated (A- ) in the aqueous solution at pH = 4.00 needs to be determined.
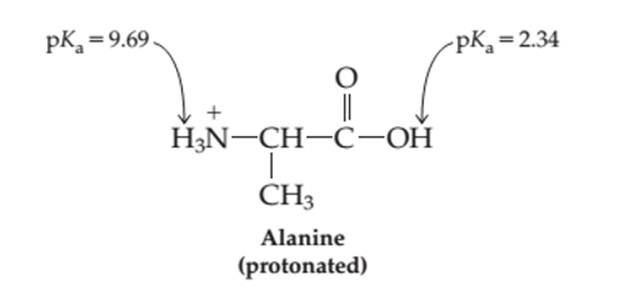
Concept introduction:
Proteins are biomolecules that are composed of certain monomer units such as amino acids. These are carboxylic acids with one amino group bonded on alpha-C atom of the molecule. These amino acids involve in condensation process to form peptides and polypeptides which further form complex protein molecules. Amino acids are the organic molecules with both
(c)
Interpretation:
The percentage of each form-protonates (H2A+ ), neutral (HA) and deprotonated (A- ) in the aqueous solution at pH = 8.50 needs to be determined.
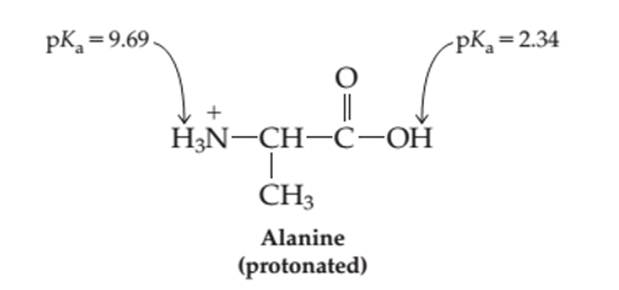
Concept introduction:
Proteins are biomolecules that are composed of certain monomer units such as amino acids. These are carboxylic acids with one amino group bonded on alpha-C atom of the molecule. These amino acids involve in condensation process to form peptides and polypeptides which further form complex protein molecules. Amino acids are the organic molecules with both
(d)
Interpretation:
The pH at which the neutral form of alanine present in the maximum amount needs to be determined.
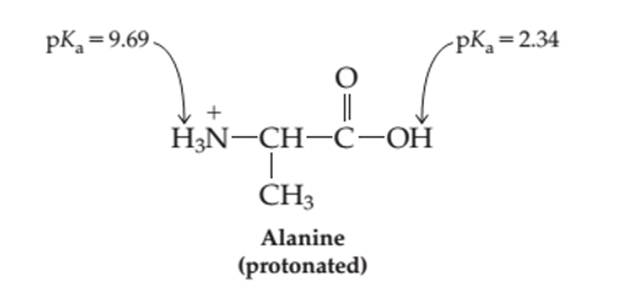
Concept introduction:
Proteins are biomolecules that are composed of certain monomer units such as amino acid. These are carboxylic acids with one amino group bonded on the alpha-C atom of the molecule. These amino acids involve in condensation process to form peptides and polypeptides which further form complex protein molecules. Amino acids are the organic molecules with both
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
EBK CHEMISTRY
- Find the value of Kb for the conjugate base of the following organic acids. (a) picric acid used in the manufacture of explosives; Ka = 0.16 (b) trichloroacetic acid used in the treatment of warts; Ka = 0.20arrow_forwardAcid-Base Equilibria Many factors contribute to the acidity of organic compounds. Electronegativity, resonance, induction, hybridization, aromaticity, and atomic size, all play a role. In the following comparisons, you are asked to identify the factor(s) that would be most important to analyze when predicting relative acidity, and then to predict the trend in acidity and pKa values. For each of the following pairs of compounds answer the following two multiple-choice questions. 1. What factor(s) are the most important to consider when predicting the relative acidity of the two compounds? a. Electronegativity of the atom possessing the hydrogen. b. Resonance stabilization of the anionic conjugate base. c. Inductive stabilization of the anionic conjugate base. d. Hybridization of the atom possessing the hydrogen. e. The atomic size of the atom possessing the hydrogen.arrow_forwardThe disinfectant phenol, C₆H₅OH, has a pKₐ of 10.0 in wa-ter, but 14.4 in methanol. (a) Why are the values different? (b) Is methanol is a stronger or weaker base than water? (c) Write the dissociation reaction of phenol in methanol. (d) Write an expres-sion for the autoionization constant of methanol.arrow_forward
- Calculate the pH at 25°C of a 0.64M solution of anilinium chloride C6H5NH3Cl. Note that aniline C6H5NH2 is a weak base with a pKb of 4.87.arrow_forwardConsider two acids: HCO2H (formic acid, pKa = 3.8) and pivalic acid [(CH3)3CCO2H, pKa = 5.0]. (a) Which acid has the larger Ka? (b) Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base? (d) When each acid is dissolved in water, for which acid does the equilibrium lie further to the right?arrow_forwardThe acid dissociation constant K of trimethylacetic acid (HC (CH3)₂CO₂) is 9.33 × 10¯º a Calculate the pH of a 1.1M solution of trimethylacetic acid. Round your answer to 1 decimal place. pH 0 X 5arrow_forward
- The acid dissociation constant K of trimethylacetic acid (HC(CH³)CO₂) is 9.33 × 10¯. Calculate the pH of a 1.2M solution of trimethylacetic acid. Round your answer to 1 decimal place. pH = 0arrow_forwardWhat is the conjugate acid of H2C6H7O5 -1aq2? What is its conjugate base?arrow_forwardAt what pH is the concentration of a compound, with a pKa = 3.7, 10 times greater in its acidic form than in its basic form?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





