
Concept explainers
(a)
Interpretation:
The possibility of resonance structure of the given structure is to be discussed.
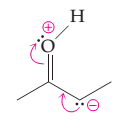
Concept introduction:
The resonance or mesmeric effect is the phenomena of delocalization of (-bonds in the chain and ring.
(b)
Interpretation:
The possibility of resonance structure of the given structure is to be discussed.
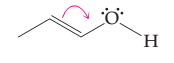
Concept introduction:
The resonance or mesmeric effect is the phenomena of delocalization of (-bonds in the chain and ring.
(c)
Interpretation:
The possibility of resonance structure of the given structure is to be discussed.
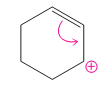
Concept introduction:
The resonance or mesmeric effect is the phenomena of delocalization of (-bonds in the chain and ring.
(d)
Interpretation:
The possibility of resonance structure of the given structure is to be discussed.
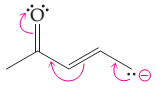
Concept introduction:
The resonance or mesomeric effect is the phenomena of delocalization of (-bonds in the chain and ring.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
EBK CHEMISTRY
- In the Lewis structure for chloromethane, the chlorine atom is sharing _____ electron pair and “owns” _____ of those electrons. Also, the chlorine atom possesses two electrons from each of _____ unshared pairs. The total number of electrons that belong to chlorine is 7 . Chlorine is a Group ____ element. The formal charge on chlorine in chloromethane is ____.arrow_forwardWhich statements are true about electronegativity? (a) Electronegativity increases from left to right in a period of the Periodic Table. (b) Electronegativity increases from top to bottom in a column of the Periodic Table. (c) Hydrogen, the element with the lowest atomic number, has the smallest electronegativity. (d) The higher the atomic number of an element, the greater its electronegativity.arrow_forwardWrite the Lewis structure for nitrosyl fluoride, FNO. Using only a periodic table, identify (a) which is the longer bond. (b) which is the stronger bond. (c) which is the more polar bond.arrow_forward
- • define electronegativity and state how electronegativity varies with position in the periodic table.arrow_forwardConsider the resonance structures of formate. Select the true statements about the resonance structures. Each carbon-oxygen bond is somewhere between a single and double bond. The actual structure of formate is an average of the two resonance forms. Each oxygen atom has a double bond 50% of the time. The actual structure of formate switches back and forth between the two resonance forms.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





