
Concept explainers
(a)
Interpretation:
The curved arrows to convert the first structure to another structure needs to be drawn as given below:
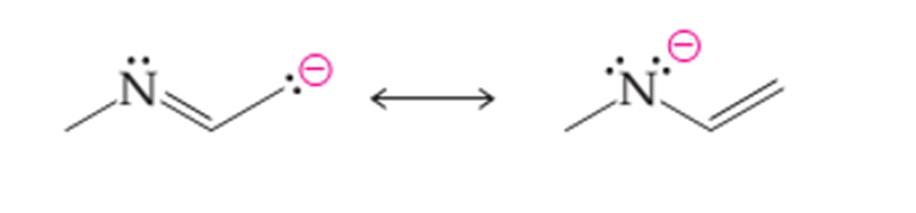
Concept introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with the parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In
(b)
Interpretation:
The curved arrows to convert the first structure to another structure needs to be drawn.
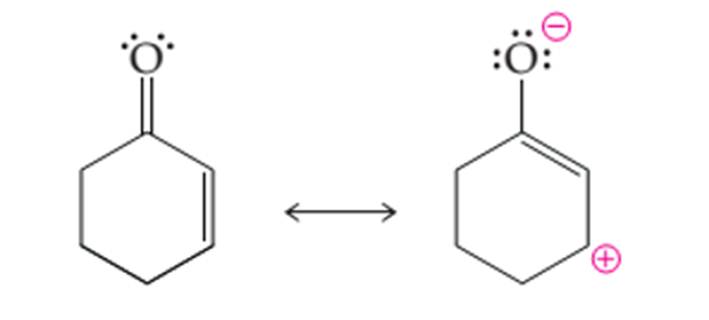
Concept introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with the parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
EBK CHEMISTRY
- The following compounds a) are NOT resonance structures, they aredifferent compounds. b) The following compound is aromaticarrow_forwardProvide the two resonance structures of the molecules shown. Circle the best resonance structure out of the two.arrow_forwardUse curved arrow notation to show how the first resonance structure can be converted to the second.arrow_forward
- Two major resonance structures are possible for the anion shown. One resonance form is given, but it is incomplete. Complete the given structure by adding nonbonding electrons and formal charges. Draw the remaining structure, including nonbonding electrons and formal charges. Omit curved arrows. Structure A: complete the structure by adding nonbonding electrons and formal charges. H H H I Structure B: draw the remaining resonance structure, including nonbonding electrons and formal charges. H- : z: H Harrow_forwardDraw the most stable resonance structure for SPC-. Explain your reasoning for why this would be the most stable.arrow_forwardThe following structure is an anion with three possible resonance contributors. One incomplete resonance form is shown below. Complete the given structure by adding nonbonding electrons and formal charges. Draw the two remaining resonance structures (in any order), including nonbonding electrons and formal charges.arrow_forward
- Draw two additional resonance structures of the following. Remember to include all formal charges needed. Clearly show the arrows to go from one structure to the next.arrow_forwardDraw the contributing resonance structures for the following. ЕНarrow_forward2) Draw the Lewis structures first and then complete the following acid-base reaction by using appropriate arrows. Acid and base should be labeled. CH3CHBRCOOH + NaHarrow_forward
- Draw all the resonance structures for periodate ion.arrow_forward22. Draw two additional resonance structures for the following compound.arrow_forwardUsing correct arrow formalism, write the contributors to the resonance hybrid structure of the acetate ion, CH3CO2, and Indicate any formal charges.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


