
Concept explainers
Interpretation:
The number of electros in the
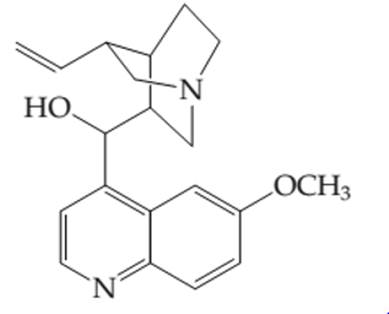
Concept introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
EBK CHEMISTRY
- Classify the compounds shown as aromatic, antiaromatic, or nonaromatic . Assume all the structures are planar.arrow_forward(1) What is the valence electron configuration for the carbon atom? (2) What is the valence electron configuration for the carbon atom?arrow_forwardUse the planar compounds below to fill in the blanks with the correct corresponding Roman numeral. II III Compound is aromatic. Compound is non-aromatic. Compound is antiaromatic.arrow_forward
- Consider the following compounds, which is aromatic?arrow_forwardDrug molecules Vicodin and Heroin, contain an amine functional group and several rings. Indicate the number of aromatic rings in each structure, and identify the amine functional group in each molecule as primary, secondary, or tertiary.arrow_forwardWrite the chemical formula, bond-line structure, and condensed formula of undecanoic acid, and draw its bond line structure.arrow_forward
- If NaCN were reacted with the given molecule, what products could the reaction form?arrow_forwardThe following compounds a) are NOT resonance structures, they aredifferent compounds. b) The following compound is aromaticarrow_forwardWhat is Resonance Theory? Sate five conclusions that can be drawn from the theory. State the two main experiments that were used to establish the extra stability of the benzene molecule. What are the factors that confer Aromaticity to an organic molecule?arrow_forward
- what is the reading of this buret?arrow_forwardDraw all aromatic hydrocarbons that have molecular formula C8H10. For each compound, determine how many isomers of molecular formula C8H9Br would be formed if one H atom on the benzene ring were replaced by a Br atom.arrow_forwardRank the compounds according to increasing positive character of the carbon atom.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



