
Concept explainers
(a)
Interpretation:
The line drawing nitrogen containing compound is to be drawn and formal charge on each atom is to be shown.
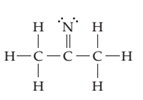
Concept introduction:
The line drawing is a chemical structure in which carbon and hydrogen molecules are not drawn. In these types of structures, lines are used to draw structure representing molecules. In the electron dot structure, the lone pair of electrons is represented as two dots on the respective symbol of the atom.
Formal charge is the charge present on each atom in the molecule; it is calculated by the following formula:
Formal Charge = Number of valence electrons on
(b)
Interpretation:
The line drawing containing compound is to drawn and formal charge on each atom is to be shown.
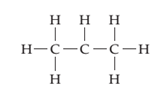
Concept introduction:
The line drawing is a chemical structure in which carbon and hydrogen molecules are not drawn. In these types of structures, lines are used to draw structure representing molecules. In the electron dot structure, the lone pair of electrons is represented as two dots on the respective symbol of the atom.
Formal charge is the charge present on each atom in the molecule; it is calculated by the following formula:
Formal Charge = Number of valence electrons on atom − [Number of non-bonded electrons + Number of bonds].
(c)
Interpretation:
The line drawing nitrogen containing compound is to drawn and formal charge on each atom is to be shown.
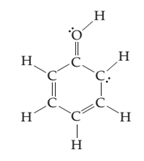
Concept introduction:
The line drawing is a chemical structure in which carbon and hydrogen molecules are not drawn. In these types of structures, lines are used to draw structure representing molecules. In the electron dot structure, the lone pair of electrons is represented as two dots on the respective symbol of the atom.
Formal charge is the charge present on each atom in the molecule; it is calculated by the following formula:
Formal Charge = Number of valence electrons on atom − [Number of non-bonded electrons + Number of bonds].
Trending nowThis is a popular solution!

Chapter 23 Solutions
CHEMISTRY-W/MASTERING CHEMISTRY ACCESS
- The Lewis structure of acetone is Circling the carbonyl carbon, i.e., the carbon atom attached to oxygen, and its octet gives Circling the oxygen atom and its octet gives Thus, atoms share electrons in making bonds, and a pair of electrons may be included in the octet of two different atoms. When computing the formal charge on an atom, the number of electrons that belong to that atom is compared with the number of electrons the atom would have in the unbonded and neutral state. If the two numbers are the same, the formal charge on the atom is zero. In a Lewis structure both electrons in an unshared pair belong to the atom, and one of every pair of shared (bonding) electrons belongs to the atom.arrow_forwardthe formal charges on all the atoms in the following Lewis diagrams. Which one would best represent bonding in the molecule Cl2O ?arrow_forwardDraw the Lewis structure created by the curved arrow. Write in formal charges wherever they are not equal to zero. H C=arrow_forward
- Provide the formal charges for each atom in the molecule below, and answer the additional question (hint: none violate the octet rule). Format your answer as +2 or -3, for example. If there is no formal charge, then enter a zer SECIN S: s this molecule have an overall charge (yes or no)? harrow_forwardDraw the Lewis structures for the following four molecules, being sure to show all steps following the methods covered in class. Structures without work shown will be marked incorrect. Also, one of these molecules has resonance structures – for this compound, make sure to include all resonance structures, indicate formal charges for each atom. SO2 OF2 IF3 NH4+ Consider a molecule where the central atom has one lone pair of electrons and is double-bonded to two other atoms (of a different element). Draw a general diagram of the molecule. Is this molecule likely a polar or nonpolar molecule? Briefly explain your reasoning, using words and the diagram.arrow_forwardDetermine the Lewis structure of each of the following polyatomic ions. Include all resonance structures (do not add any arrows between structures) and formal charges, where appropriate:arrow_forward
- Keeping the same atomic connections and moving only electrons, write a more stable Lewis structure for each of the following. Be sure to specify formal charges, if any, in the new structure. (g) (h) (i)arrow_forwardIf the atom "X" is in the fifth period of the periodic table, identify the element. Be sure toclearly explain your answer. Note: Ignore the positive formal charge on the X. Please answer the question in the image. Thank you :)arrow_forward(True/False) The summation of the formal charges of the atoms in a compound/ polyatomic ion is equal to the charge of the whole compound/ polyatomic ion.arrow_forward
- Write the resonance structure that would result from moving the electrons as the curved arrows indicate. Be sure to include formal charges if needed.arrow_forwardWhich of the ions gives a formal charge that is an accurate picture of where the charge really is?arrow_forwardDraw the number of resonance structures for SCN- . (C is the central atom; draw 3 resonance structures) Show nonzero formal charges.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning



