
Concept explainers
(a)
Interpretation:
Synthesis of benzylamine from the given starting material has to be shown.
Concept Introduction:
Preparation of imine:
An imine is a compound having
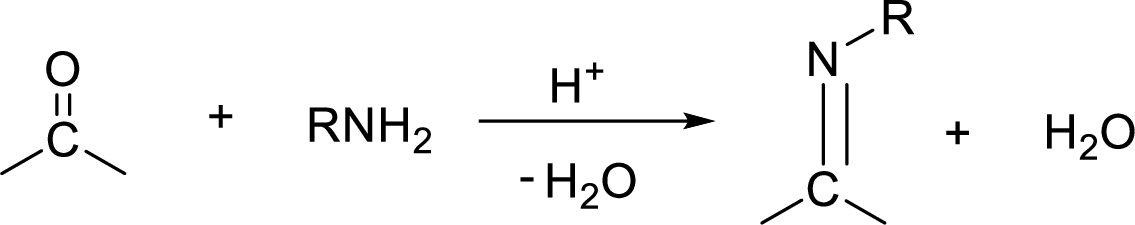
Reduction: If electrons are gained to a species or hydrogen atoms are added to a species or oxygen atom gets removed from a species during a
(b)
Interpretation:
Synthesis of benzylamine from the given starting material has to be shown.
Concept Introduction:
Amide Hydrolysis: In presence of base, amide reacts with water to form the corresponding amine and
(c)
Interpretation:
Synthesis of benzylamine from the given starting material has to be shown.
Concept Introduction:
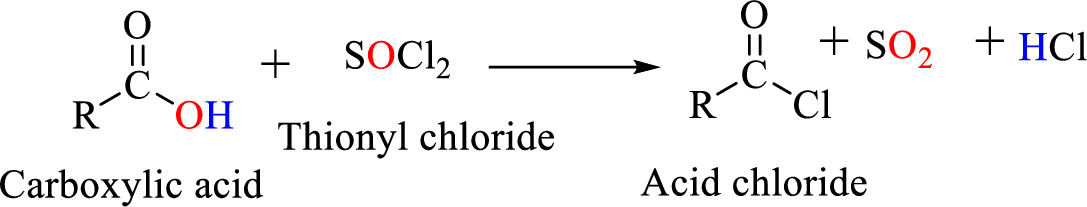
Preparation of amine: A primary amine is formed when an
Thionyl chloride:
(d)
Interpretation:
Synthesis of benzylamine from the given starting material has to be shown.
Concept Introduction:
Thionyl chloride:
(e)
Interpretation:
Synthesis of benzylamine from the given starting material has to be shown.
Concept Introduction:
Acid chlorides are most often prepared by treating a carboxylic acid with thionyl chloride.
Amide Formation: Amide is formed when an acid chloride reacts with an amine or ammonia.
Here, the chlorine atom that is attached to the carbonyl carbon atom of the acid chloride is being replaced by
Reduction: If electrons are gained to a species or hydrogen atoms are added to a species or oxygen atom gets removed from a species during a chemical reaction is known as reduction. In a reaction,
(f)
Interpretation:
Synthesis of benzylamine from the given starting material has to be shown.
Concept Introduction:
Preparation of amide: An amide is formed when an ester is reacted with ammonia.
Reduction: If electrons are gained to a species or hydrogen atoms are added to a species or oxygen atom gets removed from a species during a chemical reaction is known as reduction. In a reaction,
Trending nowThis is a popular solution!

Chapter 23 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- b) The Wolf-Kishner reduction is a reaction used in Organic Chemistry to convert carbonyl functionalities into methylene group. The reaction was used to convert an aldehyde or ketone to an alkane using hydrazine, base and thermal conditions. The mechanism begins with the attack of hydrazine of the aldehyde or ketone. Stage 1: The reaction of aldehyde/ketone with hydrazine to produce hydrazine Stage 2: Reaction with the base and heat to convert hydrozone to alkane Write the mechanism of the reaction.arrow_forwardW X Select reagent W and Y 1 HNO3 H₂SO4 2 CH3C1₂ AICI 3 3 Cl₂, FeCl3 ) 4 503, H₂SO4 NO₂ S0₂tarrow_forwardShow how to bring about each conversion in good yield.arrow_forward
- Convert phenylacetylene to butyl benzene using any necessary organic and inorganic reagents. It will require multiple steps.arrow_forwardShow how to convert 4-methoxybenzyl chloride to each amine.arrow_forwardPropose a synthesis of triphenylmethane from benzene, the only source of aromatic rings, and any other necessary reagents.arrow_forward
- Using your reaction roadmap as a guide, show how to convert cyclohexane into hexanedial. Show all reagents and all molecules synthesized along the way.arrow_forwardEthylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forwardUsing your reaction roadmap as a guide, show how to convert butane into butanal. Show all reagents needed and all molecules synthesized along the way.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


