
(a)
Interpretation:
The synthesis is to be shown for the 4-chloro-3-nitrobenzoic acid from toluene.
Concept introduction:
Nitration: The formation of nitro group in a
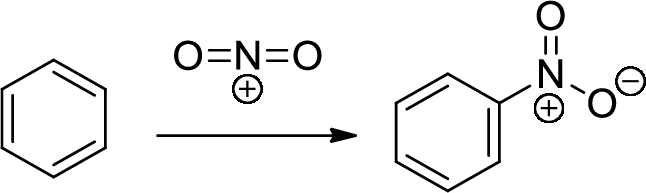
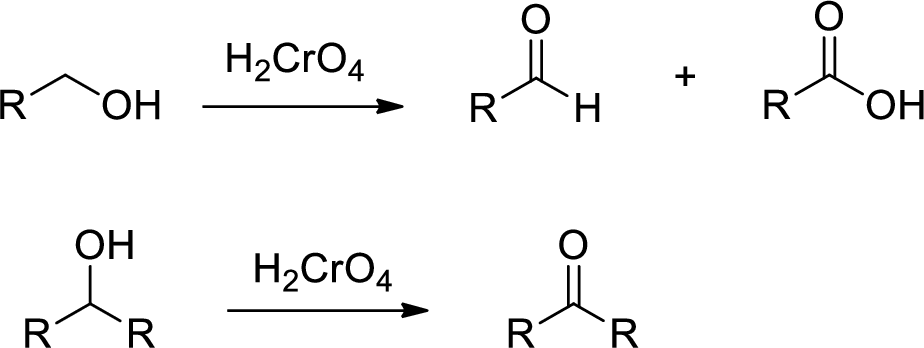
Chromic Acid:
Chromic Acid (
(b)
Interpretation:
The reagent and condition is to be proposed for step 1.
(c)
Interpretation:
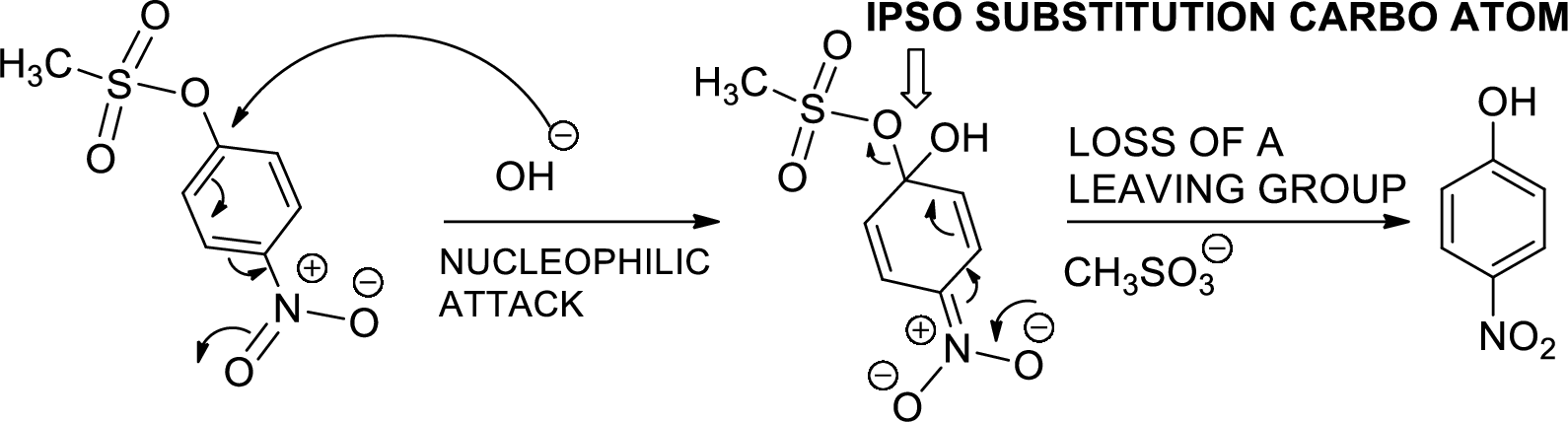
The mechanism is to be proposed for step 2.
Concept introduction:
Ipso substitution reaction: It is the one of the
(d)
Interpretation:
The reagent and condition is to be proposed for step 3.
Concept introduction:
Hydrogenolysis:
Metal catalyst gives the corresponding amine or alcohol.
Reductive amination reaction: Amination is the process by which an amine group is introduced into an organic molecule.
The conversion of Carbonyl group in to amine via imine intermediate is called reductive amination.
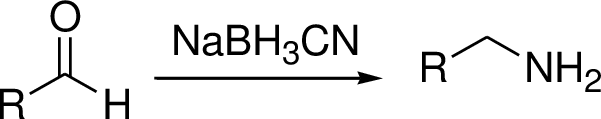
(e)
Interpretation:
The possible stereoisomer’s has to be shown if the product is chiral.
Concept introduction:
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
Trending nowThis is a popular solution!

Chapter 23 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- (b) Propose a synthesis of (2,2-dimethylpropyl)benzene from benzene.arrow_forwardShow how to synthesize the following amines from the indicated starting materials byacylation–reduction.(a) N-butylpiperidine from piperidinearrow_forwardGive the products expected when acetic formic anhydride reacts with (i) aniline and(ii) benzyl alcohol.(b) Propose mechanisms for these reactions.arrow_forward
- Propose the synthesis for the following transformations.arrow_forwardShow how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) p-aminobenzoic acid (b) 2-methyl-1-phenylbutan-2-ol (c) 5-chloro-2-methylanilinearrow_forwardIn an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?arrow_forward
- gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.arrow_forwardPropose a synthesis for propoxyphene from 1-phenyl-1-propanone and any other necessary reagents.arrow_forwardShow how you would synthesize each compound, starting with an ester containing nomore than eight carbon atoms. Any other necessary reagents may be used.(a) Ph3C¬OH (b) (PhCH2)2CHOH (c) PhCONHCH2CH3(d) Ph2CHOH (e) PhCH2OH (f) PhCOOH(g) PhCH2COOCH(CH3)2 PhCH2 C(CH2CH3)2OH(h) (i) HO¬(CH2) 8¬OHarrow_forward
- Show how you would synthesize each compound, starting with an ester containing nomore than eight carbon atoms. Any other necessary reagents may be used.(a) Ph3C¬OH (b) (PhCH2)2CHOHarrow_forwardSuggest possible synthetic route for following transformations. Show the reagents, intermediates and products.arrow_forwardSuggest how you would synthesize each compound, use cyclopentanone as one of the reagentsarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
