
Concept explainers
(a)
Interpretation:
To determine the magnetic property of the given compounds.
Concept Introduction:
Spectrochemical series: The list of ligands arranged in an ascending order of
Crystal field splitting: The energy gap between the splitting of d-orbitals of the metal ion in presence of ligands is known as the crystal field splitting
To Identify: To determine the magnetic property of the given compounds.
(a)
Answer to Problem 23.76QP
The
Explanation of Solution
Interpret the given information.
The
(b)
Interpretation:
To determine the magnetic property of the given compounds.
Concept Introduction:
Spectrochemical series: The list of ligands arranged in an ascending order of
Crystal field splitting: The energy gap between the splitting of d-orbitals of the metal ion in presence of ligands is known as the crystal field splitting
To Identify: To determine the magnetic property of the given compounds.
(b)
Answer to Problem 23.76QP
The systematic name of the given compound
Explanation of Solution
Interpret the given information.
The systematic name of the given compound
The six ligands around the metal ion give octahedral geometry.
Magnetic property of the compound:
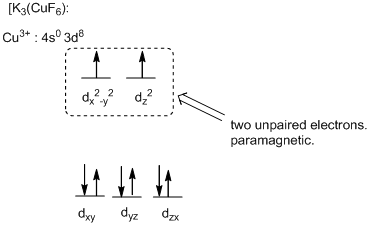
The high field complex ion possesses eight d-electrons and the two electrons are unpaired and exhibits paramagnetic.
(c)
Interpretation:
To determine the magnetic property of the given compounds.
Concept Introduction:
Spectrochemical series: The list of ligands arranged in an ascending order of
Crystal field splitting: The energy gap between the splitting of d-orbitals of the metal ion in presence of ligands is known as the crystal field splitting
To Identify: To determine the magnetic property of the given compounds.
(c)
Answer to Problem 23.76QP
The square-planar geometry will be diamagnetic in nature.
Explanation of Solution
Interpret the given information.
In complex ion of
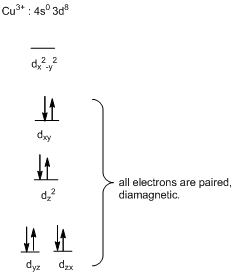
In complex ion of
Want to see more full solutions like this?
Chapter 23 Solutions
CHEMISTRY-ALEK 360 ACCES 1 SEMESTER ONL
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





