
Concept explainers
Interpretation:
The products obtained from the following reactions are to be determined: treating tyrosine with excess bromine water, reaction of phenylalanine with ethanol in the presence of hydrogen chloride, and the reaction of alanine and benzoyl chloride in aqueous base.
Concept introduction:
Amino acids contain both carboxyl group and an amino group. Both, carboxyl groups and amino groups donate and accept protons from water, respectively, to form ions. Amino acids form double ionized species by this exchange of protons in a solution between carboxyl group and amino group. These double ionized species are termed as zwitter ions.
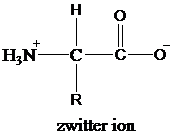
Deactivating groups like
Amino acids reacts with alcohols to produce esters, and follow nucleophilic aryl substitution reaction and the process is called Fischer esterification.
Amino acids react with acid chlorides to form acetylation or benzoylation of amino acids occur.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
- Draw the structure of the predominant form of a mixture of alanine, lysine, and aspartic acid at (i) pH 6; (ii) pH 11; (iii) pH 2.arrow_forwardDraw the structure of the phenylthiohydantoin derivatives of(a) alanine.arrow_forwardPredict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalyst (c) Br2 in CCl4arrow_forward
- Draw zwitterion forms of these amino acids. (a) Valine (b) Phenylalanine (c) Glutaminearrow_forward(a) Predict the product of the reaction of KOH with 1-amino propane. (b) Predict the product of a deprotonated ethanol (an “ethanolate anion", O-CH2-CH3) with phenol (hydroxybenzene). (c) Predict the product of propanoic acid with deprotonated ethanol (an “ethanolate anion", O-CH2-CH3).arrow_forwardh) Specify the absolute (R/S) configuration of the amino group in structure IV. (i) If the substituents in structures I, IV and V were identical (all OH or all NH2), which structure would result in a meso compound? (j) If each hydroxy group for structures I, II and VI were replaced with another amino group, which compound would be made optically inactive?arrow_forward
- Draw structural formulas for the following compounds: (a) Tributylamine(b) N-methylanilinium chloride(c) ethylammonium chloride (d) 2-amino-1-pentanol(e) 3,3-dimethyl-N-phenyl-2-aminohexane (f) 1,2-ethanediaminearrow_forwardAnother method to form a peptide bond involves a two-step process Conversion of a Boc-protected amino acid to a p-nitrophenyl ester. Why does a p-nitrophenyl ester “activate” the carboxy group of thefirst amino acid to amide formation?arrow_forwardPhenylalanine as N-termminus is incorrect. Please provide the correct solution.arrow_forward
- 22-9 What is the difference in structure between tyrosine and phenylalanine?arrow_forward22-20 Show how alanine, in solution at its isoelectric point, acts as a buffer (write equations to show why the pH does not change much if we add an acid or a base).arrow_forwardA chemically modified guanidino group is present in cimetidine (Tagamet), a widely prescribed drug for the control of gastric acidity and peptic ulcers. Cimetidine reduces gastric acid secretion by inhibiting the interaction of histamine with gastric H2 receptors. In the development of this drug, a cyano group was added to the substituted guanidino group to alter its basicity. Do you expect this modified guanidino group to be more basic or less basic than the guanidino group of arginine? Explain.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT




