
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 27, Problem 27.39P
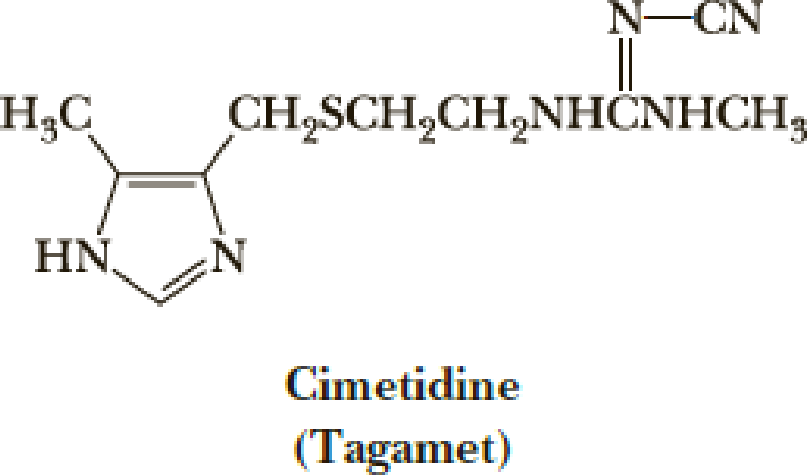
A chemically modified guanidino group is present in cimetidine (Tagamet), a widely prescribed drug for the control of gastric acidity and peptic ulcers. Cimetidine reduces gastric acid secretion by inhibiting the interaction of histamine with gastric H2 receptors. In the development of this drug, a cyano group was added to the substituted guanidino group to alter its basicity. Do you expect this modified guanidino group to be more basic or less basic than the guanidino group of arginine? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Carnosine, found in muscle and brain tissue, acts as a buffer to neutralize small amounts of acid. The pKa of the conjugate acid of carnosine is close to 7.0. What is its structure?
A naturally occurring amino acid such as alanine has a group that is a carboxylic acid and a group that is a protonated amine. a. If the pKa value of a carboxylic acid such as acetic acid is about 5 (see Table 2.1), then why is the pKa value of the carboxylic acid group of alanine so much lower? b. Draw the structure of alanine in a solution at pH = 0. c. Draw the structure of alanine in a solution at physiological pH (pH 7.4). d. Draw the structure of alanine in a solution at pH = 12. e. Is there a pH at which alanine is uncharged (that is, neither group has a charge)? f. At what pH does alanine have no net charge (that is, the amount of negative charge is the same as the amount of positive charge)?
Treating chitin with H2O, -OH hydrolyzes its amide linkages, forming a compound called chitosan. What is the structure of chitosan? Chitosan has been used in shampoos, bers for sutures, and wound dressings.
Chapter 27 Solutions
Organic Chemistry
Ch. 27.1 - Of the 20 protein-derived amino acids shown in...Ch. 27.2 - Prob. 27.2PCh. 27.2 - Prob. 27.3PCh. 27.3 - Draw a structural formula for Lys-Phe-Ala. Label...Ch. 27.4 - Which of these tripeptides are hydrolyzed by...Ch. 27.4 - Deduce the amino acid sequence of an undecapeptide...Ch. 27.6 - Prob. 27.7PCh. 27 - What amino acid does each abbreviation stand for?...Ch. 27 - The configuration of the chiral center in -amino...Ch. 27 - Assign an R or S configuration to the chiral...
Ch. 27 - Prob. 27.11PCh. 27 - Prob. 27.12PCh. 27 - Draw zwitterion forms of these amino acids. (a)...Ch. 27 - Prob. 27.14PCh. 27 - Why is Arg often referred to as a basic amino...Ch. 27 - Prob. 27.16PCh. 27 - Prob. 27.17PCh. 27 - Prob. 27.18PCh. 27 - Prob. 27.19PCh. 27 - Prob. 27.20PCh. 27 - Both norepinephrine and epinephrine are...Ch. 27 - Prob. 27.22PCh. 27 - Draw a structural formula for the form of each...Ch. 27 - Prob. 27.24PCh. 27 - Write the zwitterion form of alanine and show its...Ch. 27 - Prob. 27.26PCh. 27 - Write the form of aspartic acid most prevalent at...Ch. 27 - Prob. 27.28PCh. 27 - Prob. 27.29PCh. 27 - For lysine and arginine, the isoelectric point,...Ch. 27 - Prob. 27.31PCh. 27 - Account for the fact that the isoelectric point of...Ch. 27 - Prob. 27.33PCh. 27 - Prob. 27.34PCh. 27 - At pH 7.4, the pH of blood plasma, do the majority...Ch. 27 - Prob. 27.36PCh. 27 - Prob. 27.37PCh. 27 - Prob. 27.38PCh. 27 - A chemically modified guanidino group is present...Ch. 27 - Draw a structural formula for the product formed...Ch. 27 - Prob. 27.41PCh. 27 - Prob. 27.42PCh. 27 - A decapeptide has the following amino acid...Ch. 27 - Following is the primary structure of glucagon, a...Ch. 27 - Prob. 27.45PCh. 27 - Draw a structural formula of these tripeptides....Ch. 27 - Estimate the pI of each tripeptide in Problem...Ch. 27 - Glutathione (G-SH), one of the most common...Ch. 27 - Following are a structural formula and a...Ch. 27 - Prob. 27.50PCh. 27 - Prob. 27.51PCh. 27 - Prob. 27.52PCh. 27 - Prob. 27.53PCh. 27 - Prob. 27.54PCh. 27 - Distinguish between intermolecular and...Ch. 27 - Prob. 27.56PCh. 27 - Prob. 27.57P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- fid the molecular structure of 1. deprotonated form of 2-amino-3hydroxypropanoic acid 2.zwitterionic form of 2-Amino-3-methylpentanoic acid 3. protonated form of 2-Amino-3-methylpentanoic acid 4. deprotonated form of 2,5 diamino5-oxopentanoic acid 5. zwitterionic form of 2-amino-3hydroxypropanoic acid 6.2-phenyl propanol 7. 2,4,6-trinitrophenol 8. oxoheptanoic acid 9. (4E)-2,6-dimethylhepta2,4-dien3ol 10. 3-methylbutan1-olarrow_forwardGive the following1. an amino acid that has an aryl side group chain 2. an amino acid that contains a side chain with a thioether bond3. an amino acid that reacts with HCL through acid-base reaction4. an amino acid that contains a sulfhydryl side chain5. an amino acid with a side chain that can form hydrogen bonds with water6. an amino acid that contains an amide side chainarrow_forwardTyrosine is an amino acid whose side chain has a pKa of 10.1. At pH 7, what protonation form would you expect to find it in?arrow_forward
- The side chain of acetyl-L-lysine-OMe has a pKa=10.5 and its acid-dissociation reaction is shown below. C. what is the pH of a solution wherein the ration of [A-]/[HA]=1:10?arrow_forwardEnzyme-catalyzed decarboxylation of glutamic acid gives 4-aminobutanoic acid . Estimate the pI of 4-aminobutanoic acidarrow_forwardValine has pKa's of 2.286 and 9.719. Estimate the fractional composition of Valine in the -1 form at pH=6.arrow_forward
- α-Amino acids can be prepared by treating an aldehyde with ammonia/trace acid, followed by hydrogen cyanide, followed by acid-catalyzed hydrolysis. What amino acid is formed when the aldehyde that is used is 3-methylbutanal?arrow_forwardOrnithine is an amino acid that is not used in the synthesis of proteins, but is an important intermediate in several metabloic pathways including the urea cycle and the synthesis of polyamines. It has a perfectly ordinary terminal amino group and terminal carboxyl group like any other amino acid (so use the pKas for those groups given on your amino acids handout), and a side chain with a single ionizable side group with a pKa of 10.3. If ornithine is placed in solution at pH 7.0, it has a net charge of +1. What would the net charge on this amino acid be if the pH of the solution was raised to pH 12.0? Please explain your reasoning.arrow_forward(b) Describe how the charge of some amino groups in a protein might differ at pH 9.0 and pH 5.0. the charge on the amino group will differ at pH 5 and pH 9 which will depend on the pKa of the amino acid. (c) Describe how the charge of some carboxyl groups in a protein might differ at pH 9.0 and pH 5.0. charge on carboxyl group will differ at pH 5 and pH 9 which will depend on the pKa of the amino acid. (d) Given your answers to parts (b) and (c), what kind of intramolecular interactions in beta-galactosidase are most likely to be affected by a change in pH from 9.0 to 5.0? (e) Could the interactions you mention in part (d) affect the catalytic activity of beta-galactosidase?arrow_forward
- Is TMA a volatile compound or a biogenic amine? Is measurement of TMA a reliable index of spoilage? Why and why not?arrow_forwardThe imidazole ring on Histamine has a pKa of 6.5. What is the %ionization of this group at pH 8.2?arrow_forwardThe shown image is a reaction diagram for the hydrolysis of peptide bonds. Appropriately substituted phosphonates (structure shown above) were successfully designed as strong inhibitors for proteolytic enzymes. Explain why this strategy was successful. Note that the lengths of P–O bonds in phosphonates are significantly longer than the corresponding C–O bonds.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY