
Concept explainers
(a)
Interpretation:
The structures of testosterone and estradiol has to be compared.
Concept introduction: Steroids are an example of lipids. They are biologically active organic compounds and are insoluble in water and soluble organic solvents. Example of steroids includes testosterone, estradiol, and cholesterol.
All steroids have common
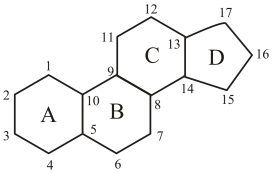
(b)
Interpretation:
The given two structures has to be explained as steroids.
Concept introduction: Steroids are an example of lipids. They are biologically active organic compounds and are insoluble in water and soluble organic solvents. Example of steroids includes testosterone, estradiol, and cholesterol.
All steroids have common

Trending nowThis is a popular solution!

Chapter 24 Solutions
Chemistry & Chemical Reactivity
- Write the full reaction for the amidation of acetic acid and ammonia Was there a similar reaction between aniline and ammonia Explain.arrow_forwardSuppose you were given the structural formula of compound B but only the molecular formulas for compound A and the starting bicyclic amine. Given this information, is it possible, working backward, to arrive at an unambiguous structural formula for compound A? for the bicyclic amine?arrow_forwardWould you expect the following compound to act as a soap? Why or why not? We explain in Chapter 3 that a chaincomposed of multiple, adjacent, single bonds is flexible. How does this new information affect your answer?arrow_forward
- Give the chemical names of the corresponding structures for compounds A and B.arrow_forwardWhat reagents would you use to chemically extract benzoic acid and valeric acid? Or can it not be chemically extracted? The image shows the compound structure.arrow_forwardWrite the products of the reaction of diphenhydramine (a base) with the acid HCl shown below. Are the reactants or products more soluble in water? Briefly explain.arrow_forward
- Predict the same for a) methyl propanoate and b) propanamidearrow_forwardThe compound acetophenone has a very similar molar mass to that of benzoic acid and benzamide. However, acetophenone has a much lower m.p. (20 °C) than both such that, by contrast, it is a liquid at room temperature. By considering intermolecular forces and comparing functional group structure, account for this big difference in physical properties.arrow_forward(b) Which of the following common amino acids has more than one amino group?arrow_forward
- Provide a structurearrow_forwardWhen the phenol red dye has turned yellow in solution, would phenol red be in the predominate ionized or un-ionized form?arrow_forwardExplain which amide (A, B or C) would be mainly found in the hexane layer, if there are all added to a water/hexane mixture?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


