
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 24, Problem 24.16P
Interpretation Introduction
Interpretation:
Structural formula for palladium-containing intermediates in the given Heck reaction has to be proposed.
Concept Introduction:
The
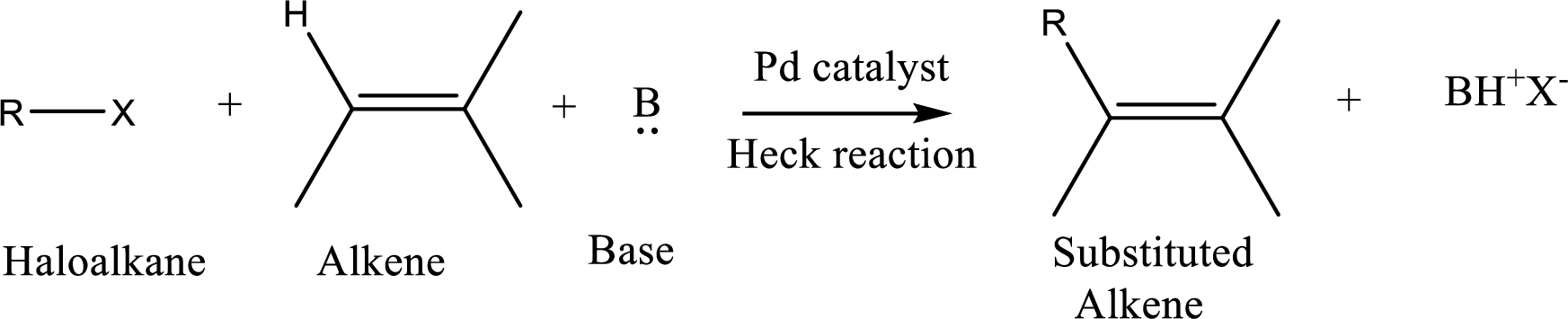
Reaction is highly regioselective in which formation of a new carbon-carbon bond occurs at a less substituted carbon of a double bond.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose an arrow-pushing mechanism for this reaction
Propose mechanisms to show the interchange of protons between ethanol molecules under(a) acid catalysis
Provide a step by step mechanism for the conversion of cuclohexanol in hot concentrated phosphoric acid
Chapter 24 Solutions
Organic Chemistry
Ch. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.4 - Prob. 24.3PCh. 24.5 - Show how the following compound can be prepared...Ch. 24.5 - Prob. 24.5PCh. 24.5 - Prob. 24.6PCh. 24.6 - Prob. 24.7PCh. 24 - Prob. 24.8PCh. 24 - Prob. 24.9PCh. 24 - Prob. 24.10P
Ch. 24 - Treatment of cyclohexene with iodobenzene under...Ch. 24 - Prob. 24.12PCh. 24 - Prob. 24.13PCh. 24 - The aryl diene undergoes sequential Heck reactions...Ch. 24 - Heck reactions take place with alkynes as well as...Ch. 24 - Prob. 24.16PCh. 24 - The following transformation involves a series of...Ch. 24 - Show the sequence of Heck reactions by which the...Ch. 24 - Prob. 24.19PCh. 24 - Write the steps that are critical in the following...Ch. 24 - Prob. 24.21PCh. 24 - Prob. 24.22PCh. 24 - Prob. 24.23PCh. 24 - Show how the following compound could be prepared...Ch. 24 - It is typically very difficult to do a...Ch. 24 - The compound eutypine is an antibacterial agent...Ch. 24 - Prob. 24.27PCh. 24 - Prob. 24.28PCh. 24 - Prob. 24.29PCh. 24 - Prob. 24.30PCh. 24 - Prob. 24.31PCh. 24 - Prob. 24.32PCh. 24 - Prob. 24.33PCh. 24 - The following transformation can be accomplished...Ch. 24 - Prob. 24.35PCh. 24 - Prob. 24.36PCh. 24 - Prob. 24.37PCh. 24 - Prob. 24.38PCh. 24 - E. J. Coreys 1964 total synthesis of...Ch. 24 - Prob. 24.40P
Knowledge Booster
Similar questions
- Heck reactions take place with alkynes as well as alkenes. The following conversion involves an intramolecular Heck reaction followed by an intermolecular Heck. Propose structural formulas for the palladium-containing intermediates involved in this reaction.arrow_forwardShow the sequence of Heck reactions by which the following conversion takes place. Note from the molecular formula given under each structural formula that this conversion corresponds to a loss of H and I from the starting material.arrow_forwardGive clear handwritten reactions of these three subparts!arrow_forward
- Show step by step reactions with proper reagents how you would carry out the following chemical conversion.arrow_forwardPropose a mechanism for the acid-catalyzed hydration of propene. Remember that a mechanism must include curved arrows to show movement of electrons, as well as all intermediates.arrow_forwardhe reaction that occurs when the benzaldehyde in your hand is reacted in a basic environment. Cannizzaro reaction is called benzoin recovery when reacted with cyanide. It Explain the reason by writing the reaction mechanisms of the reactions.arrow_forward
- In a Grignard synthesis of benzoic acid experiment, a student determines that their crude product was a mixture of benzoic acid and benzene. Describe a series of extractions using the following solvents and solutions to get pure benzoic acid or its salt in an ether solution. A. Methyl tert-butyl ether (MTBE) B. 3M HCl C. 3M NaOHarrow_forwardIn a paragraph form, provide the experimental procedures of The second stage is the 3-benzoylpropionic acid reaction of Clemmenson, which results in 4-phenyl butanoic acid. Clemmensen reaction is observed in the presence of zinc amalgam and hydrochloric acid.arrow_forwardWhen the hypochlorite oxidation is performed on secondary alcohols such as 1-phenolethanol, the product of the reaction is benzoate ion and trichloromethane if an excess of hypochlorite is used in the absence of a buffer to keep the pH below 10. Explain the formation of these products (Hint: what reaction do the products remind you of?) Provide a complete mechanism?arrow_forward
- Propose reagents and conditions for Step 1. Account for the fact that the CF3 group is meta directing.arrow_forwardThis is a transesterification reaction. It mechanistically is the same as a Fischer Esterification. Write a mechanism for the reaction. Include all lone pairs and formal charges present throughout the mechanism.arrow_forwarda)Step by step show the reaction of 1 mol of water from the 3,3-dimethyl-2-pentanol compound in acid-catalyzed medium.Name the main product formed. b)Indicate whether the resulting product shows the geometric isomer or not. c)Write the products formed when the product is ozonized and oxidized.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT