
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 24, Problem 24.30P
Interpretation Introduction
Interpretation:
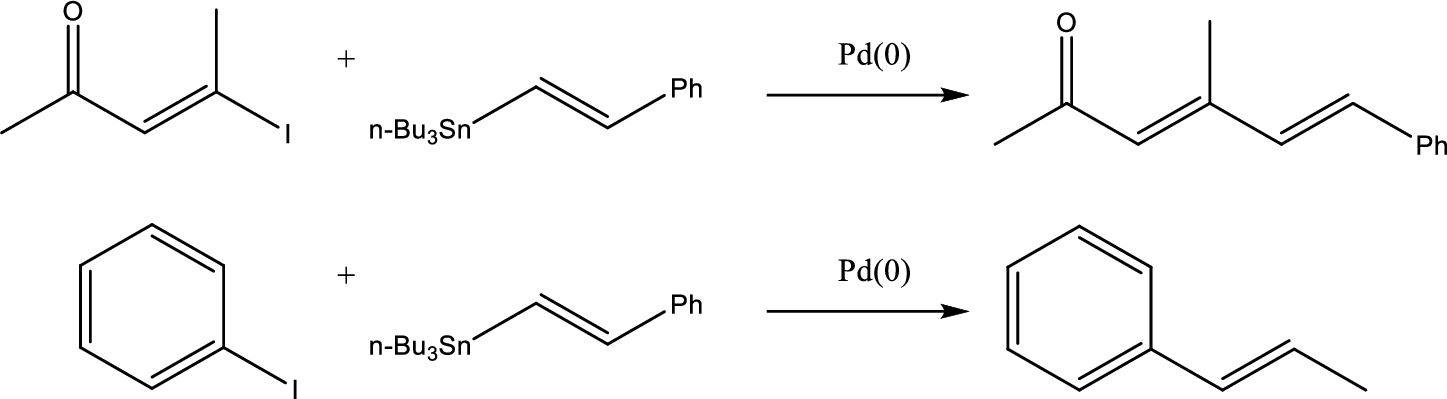
The coupling reaction and the staring material to form the given product has to be identified.
Concept Introduction:
Stille coupling uses a tin reagent (
Stille coupling gives conjugated dienes or alkenyl aryl system.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given that a primary carbocation is less stable than a tertiary carbocation, would you expect that a reaction that creates a primary carbocation would be more or less responsive to solvent polarity than a reaction that creates a tertiary carbocation, and why?
For this mechanism, why are there two E1 major products? Are both of the E1 products of equal stability? Why or why not? What is the relative stability of the cycloalkenes with methyl?
Also, why doesn't SN1 have two products? Is it because shifting the hydride would make it less stable?
Explain the following result. Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.
Chapter 24 Solutions
Organic Chemistry
Ch. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.4 - Prob. 24.3PCh. 24.5 - Show how the following compound can be prepared...Ch. 24.5 - Prob. 24.5PCh. 24.5 - Prob. 24.6PCh. 24.6 - Prob. 24.7PCh. 24 - Prob. 24.8PCh. 24 - Prob. 24.9PCh. 24 - Prob. 24.10P
Ch. 24 - Treatment of cyclohexene with iodobenzene under...Ch. 24 - Prob. 24.12PCh. 24 - Prob. 24.13PCh. 24 - The aryl diene undergoes sequential Heck reactions...Ch. 24 - Heck reactions take place with alkynes as well as...Ch. 24 - Prob. 24.16PCh. 24 - The following transformation involves a series of...Ch. 24 - Show the sequence of Heck reactions by which the...Ch. 24 - Prob. 24.19PCh. 24 - Write the steps that are critical in the following...Ch. 24 - Prob. 24.21PCh. 24 - Prob. 24.22PCh. 24 - Prob. 24.23PCh. 24 - Show how the following compound could be prepared...Ch. 24 - It is typically very difficult to do a...Ch. 24 - The compound eutypine is an antibacterial agent...Ch. 24 - Prob. 24.27PCh. 24 - Prob. 24.28PCh. 24 - Prob. 24.29PCh. 24 - Prob. 24.30PCh. 24 - Prob. 24.31PCh. 24 - Prob. 24.32PCh. 24 - Prob. 24.33PCh. 24 - The following transformation can be accomplished...Ch. 24 - Prob. 24.35PCh. 24 - Prob. 24.36PCh. 24 - Prob. 24.37PCh. 24 - Prob. 24.38PCh. 24 - E. J. Coreys 1964 total synthesis of...Ch. 24 - Prob. 24.40P
Knowledge Booster
Similar questions
- What is the answer to this reaction? (with all possible stereoisomers please!)arrow_forwardWhat is the product (include the stereochemistry) of the 6 pi electron electrocyclic reaction below, use the appropriate Woodward-Hoffman rule.arrow_forwardwhat structure show the transition state for the rate determining step in the sn1 reaction with this picture?arrow_forward
- Which of the following schemes is the best mechanism for the base-catalyzed conversion of 2-butanone to the enol tautomer shown below?arrow_forwardWhat is the best way to turn benzene into the following product? Would friedel-crafts alkylation followed by some sort of nitration work or would that be too low yield?arrow_forwardWhen carrying out this two step procedure (bromination and subsequent elimination) on cyclopentene, the lab students were surprised to find out that an alkyne was not the final product. What would the final product most likely be?arrow_forward
- Write the products of the reaction below and indicate if the product is R or S or racemic or achiral.arrow_forwardIodoethane does not react via Sn2 reaction with NaI in Acetone. It is a good leaving group and on the primary carbon, why doesn't it react Sn2? (I know it reacts Sn1) I would very much appreciate it if someone can type the answer and draw a picture of the mechanism.arrow_forwardCan you write out the mechansim showing curve arrows of this synthesis of acetylferrocenearrow_forward
- which newman projection displays the proper antiperiplanar orientation for an E2 elimination of the reaction sequence belowarrow_forwardAccording to the Woodward-Hoffmann rules, is this cycloreversion reaction possible under these conditions?arrow_forwardFill in the missing reagents or major products to complete the transformations. Be sure to pay carefulattention to stereochemistry where appropriate. If the major product is a pair of enantiomer you mayshow one of them and indicate + enantiomer. If the products are diasteromers, show them both.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
