
Organic Chemistry
8th Edition
ISBN: 9781337516402
Author: Brown
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 24.25P
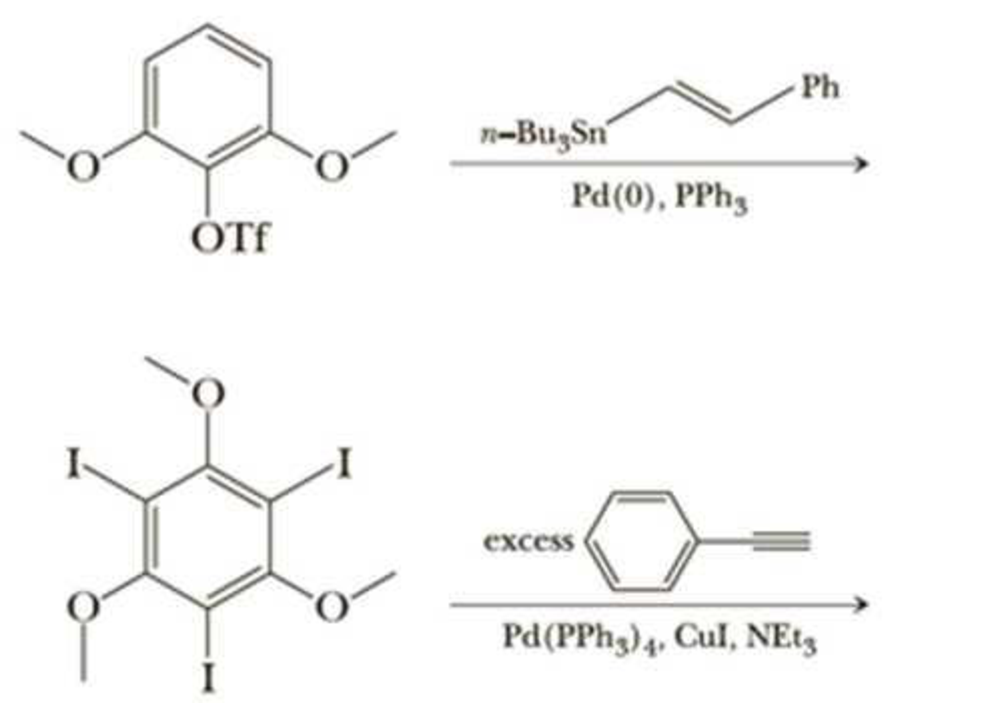
It is typically very difficult to do a substitution reaction on an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
It is typically very difficult to do a substitution reaction on an aromatic ring when the
leaving group is flanked by two other bulky substituents. Moreover, in Section 22.3, we
found that nucleophilic aromatic substitution requires strongly electron-withdrawing
groups on the benzene ring. However, Pd-catalyzed coupling allows entry into such
products. As examples, write the products of the following reactions and state which
coupling reaction is being utilized.
Ph
n-BugSn
Pd(0), PPhg
ÓTf
excess
Pd(PPH3)4, Cul, NE13
Why does the addition of bromine to fumaric acid require a high temperature for the reaction while other substrates (generic alkanes, alkenes, and alkynes) react with bromine at room temperature?
The Friedel-Crafts Alkylation of p-xylene n-propyl bromide can result in an isopropyl as well as n-propyl substitution.
A) Write the chemical equation involved in this reaction.
B) Which will be the major and minor product? What do you expect will be the ratio of n-propyl to isopropyl substitution for p-xylene?
Chapter 24 Solutions
Organic Chemistry
Ch. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.4 - Prob. 24.3PCh. 24.5 - Show how the following compound can be prepared...Ch. 24.5 - Prob. 24.5PCh. 24.5 - Prob. 24.6PCh. 24.6 - Prob. 24.7PCh. 24 - Prob. 24.8PCh. 24 - Prob. 24.9PCh. 24 - Prob. 24.10P
Ch. 24 - Treatment of cyclohexene with iodobenzene under...Ch. 24 - Prob. 24.12PCh. 24 - Prob. 24.13PCh. 24 - The aryl diene undergoes sequential Heck reactions...Ch. 24 - Heck reactions take place with alkynes as well as...Ch. 24 - Prob. 24.16PCh. 24 - The following transformation involves a series of...Ch. 24 - Show the sequence of Heck reactions by which the...Ch. 24 - Prob. 24.19PCh. 24 - Write the steps that are critical in the following...Ch. 24 - Prob. 24.21PCh. 24 - Prob. 24.22PCh. 24 - Prob. 24.23PCh. 24 - Show how the following compound could be prepared...Ch. 24 - It is typically very difficult to do a...Ch. 24 - The compound eutypine is an antibacterial agent...Ch. 24 - Prob. 24.27PCh. 24 - Prob. 24.28PCh. 24 - Prob. 24.29PCh. 24 - Prob. 24.30PCh. 24 - Prob. 24.31PCh. 24 - Prob. 24.32PCh. 24 - Prob. 24.33PCh. 24 - The following transformation can be accomplished...Ch. 24 - Prob. 24.35PCh. 24 - Prob. 24.36PCh. 24 - Prob. 24.37PCh. 24 - Prob. 24.38PCh. 24 - E. J. Coreys 1964 total synthesis of...Ch. 24 - Prob. 24.40P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The oxidation of an aldehyde is commonly carried out using potassium permanganate or potassium dichromate. In fact, the carbon alpha to a benzene ring is very reactive, hence it is possible to oxidize an alkyl benzene compound at the alpha position. Give the equation for the reaction of toluene to benzoic acid using potassium permanganate.arrow_forwardAnilines can be converted into diazonium salts by reaction with nitrous acid. Diazonium salts react with phenols to form azo dyes. Sketch the aniline derivative and phenol reactant that can combine to produce the azo dye shown.arrow_forwardIn the discussion of poly(ether ether ketone), or PEEK, in Section 26.6a.2, we observed that the addition of a tert-butyl group to the aromatic ring increases the solubility of a PEEK in organic solvents. However, when researchers added a second tert-butyl group, as shown here, they found that the solubility of the PEEK decreased. Explain this observation. A PEEK with two tert-butyl groups per repeating unitarrow_forward
- 1) Discuss three methods that can be utilised to synthesise n – heptane in the laboratory.arrow_forwardWrite a series of reaction leading to parabromoethylbenzene, beginning with benzene and using other reagents as needed. What isomeric side products might also be formed?arrow_forwardAn electron-deficient carbon atom reacts with a nucleophile, symbolized as: Nu−. Define this ?arrow_forward
- 6. Write the structure of the organic product in each of the following reactions. Assume only monosubstitution for electrophilic aromatic substitution reactions. B HNO, acetic acid OH بلی NO₂ CI NO₂ Br₂ CHC NHANH, "OH CHÍNH, H₂SOarrow_forwardWhich of the following chemical equations depicts an alkylation reaction? C6H6() + CH3Cl() → C6H5CH3() + HCl(g) 2 CH3OH() + 3 O2(g) → 2 CO2(g) + 4 H2O() C6H12() → C6H10() + H2(g) CH2ClCH2Cl(g) + H2(g) → CH3CH3(g) + Cl2(g) CHClCHCl(g) → CH2ClCH2Cl(g)arrow_forwardQuestion #19.arrow_forward
- Black powder (see introduction) was the propellant used in rifles and pistols during the 1860’s. Would it have been possible to use diphenylamine to detect gunshot residue on John Wilkes Booth after the assignation of President Lincoln? Why or Why not?arrow_forwardExplains the general concept of the manufacture of benzene by dealkylation of toluene with hydrogen.arrow_forwardDraw the structure of each of the following molecules. (a) pentanoic anhydride; (b) hexanoic propanoic anhydride; (c) ethanoic 3-methylpentanoic anhydridearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY